Monitoring Parkinson's with digital accuracy
-
Research
- Technologies for the Future
Posted on 28 March 2018
We’re developing monitors to accurately measure the progression of Parkinson’s, and the side effects of its treatment.
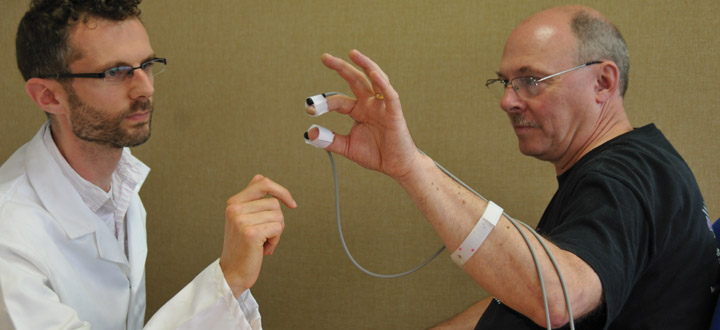
Clinicians can monitor the progress of Parkinson’s with greater accuracy using the PD-Monitor.
I would like to think that in the future, however it happens, the technology is in use and benefiting people living with the condition. If that’s the case then I think we’ve done our job, which is to take the research we’ve done and get it to the patients.
In 2015, cutting edge research at the University of York’s School of Physics, Engineering and Technology led to innovative uses of technology in improving and refining the treatment of Parkinson’s-related conditions.
Since then the work has continued, and the technology has been developed to include the Levodopa-induced Dyskinesia Monitor (LID-Monitor), enabling clinicians to accurately monitor the side effects of Parkinson’s medication, and the PD-Monitor, which gives medical staff the ability to track the progress of the condition with an extremely high degree of accuracy and uniformity.
Both innovations have the potential to greatly improve the quality of data medical staff can access when prescribing medication or assessing the progress of the condition.
Professor Stephen Smith is leading on the research, and heads ClearSky Medical Diagnostics, the company formed to market both products. His team have worked closely with consultant neurologists and specialist nurses, as well as those living with the condition, to create this unique medical equipment.
Professor Smith says: “We’ve made great progress in that both the LID and PD-Monitors now have regulatory approval. The LID-Monitor is in use in Leeds and Harrogate hospitals with York and Scarborough hospitals also about to start using them. The PD-Monitor is in use in a clinical trial in the UK to assess the effectiveness of statins in the treatment of Parkinson’s.”
LID Monitor
The condition known as Parkinson’s is caused by the dying of cells that produce the chemical dopamine, which carry the signals from the brain to various part of the body, resulting in reduced control of limbs. One way of treating this aspect of the condition is to administer artificial dopamine to restore some control to the person living with the condition.
“The trouble is after five years half of those with the condition suffer side effects and after ten years that figure rises to 90 per cent,” says Professor Smith. “The effect is uncontrolled movement, known as dyskinesia, which can cause all sorts of difficulties, including injuries from falls and a loss of balance, both for the individual and their families.”
Information
With the right level of information though, the situation can be improved, and that’s where the LID-Monitor can help.
“The idea is that the side effects can be managed if the clinicians have enough information about when they occur, how severe they are, and how the medication is affecting them. Without the LID-Monitor, in severe cases, specialist nurses would assess the patient sometimes every hour for a full 24 hours, which can be difficult, and the results of the assessment can be subjective.”
The LID-Monitor consists of six small monitors, worn by the subject in their own home for a 24-hour period. The sensors track the subject’s movement continuously throughout the day, after which the data is uploaded for analysis, and deliver an added benefit of reducing the need for the person to visit a hospital, which can be difficult for a person with Parkinson’s.
Professor Smith says: “The readings are fed through our evolutionary algorithms which are trained to recognise the difference between movement caused by side effects of the medication and those associated with ‘normal’ movement.”
Accuracy
The resulting chart clearly shows how the body is responding to the medication with a high degree of digital accuracy.
“This is an objective approach whereas conventional clinical assessment is subjective,” says Professor Smith. “So currently there’s always a level of uncertainty in making these assessments. And this test is done over 24 hours, and it’s often not practical for a specialist nurse to assess over that time.”
The accuracy of the data means the clinical staff have much more refined information with which to make decisions when adjusting medication, so the recipient is much more likely to get the amount of medication which works for them, and benefits are felt far more quickly.
“When medication is adjusted it can sometimes take months to assess whether the adjustments are working,” says Professor Smith. “So if the adjustments are correct initially, then it leads to a better result for the person living with the condition.”
PD-monitor
The PD-Monitor is another device Professor Smith’s team has produced, this time to monitor the progression of the condition with highly refined digital accuracy.
Professor Smith explains his team’s approach has always been to take a standard task, familiar to clinical staff, and digitise it, rather than inventing entirely new routines which can add to the administrative burden and require retraining.
He says: “In this case we took the ‘finger tapping test’, which is one of a number of standard tests people undertake when clinicians are assessing the progress of the condition. The subject is asked to hold their thumb and index finger wide apart and tap them together ten times and the clinician then rates the tapping on a scale of zero to four.”
The problem with this approach is that variances from practice to practice mean that the readings can be very subjective and inconsistent, particularly in the middle (two to three) range, and that’s where the PD-Monitor helps.
Before taking the test a sensor is attached to the subject’s index finger and thumb, which measures the movement of the digits in three-dimensional (3D) space. These give the medical staff highly accurate, consistent readings.
Algorithms
That data is fed through algorithms which give a far more precise reading than had previously been possible, eliminating the individual interpretation of movement, which could lead to variances in measurement.
This means that over time - assessments usually take place every six months - the progress of the condition can be measured very accurately, meaning that the 0-4 scale can be refined to a point scale, such as progression from 2.0 to 2.4 over six months, giving the subject a precise, easy to understand progress report.
“Specialists have told me that when they see a person living with the condition they might have only 15 minutes to spend with them,” says Professor Smith. “At least 10 minutes of that time can be taken up by trying to explain how the Parkinson’s has progressed in the last six months, rather than addressing the more important issues of medication and quality of life. So to be able to show people an objective report saves a lot of time and enables those living with the condition to quickly and clearly understand its progress; that was an unexpected benefit.”
And the PD-Monitor is now being included in a nationwide clinical trial to assess the effectiveness of statins in slowing the progress of the condition. The trial is being carried out in 25 hospitals across the UK and standard finger tapping tests are being used, with the monitor also being used in five of the 25 hospitals.
“They’re using our equipment alongside the existing finger tapping test method, so hopefully we can give them a much more objective and refined measure of whether the statins are slowing the condition or not,” says Professor Smith. “This is in progress, but if things go well we’ll be able to say that by using our technology the study reached a level of objectivity it would not have achieved by traditional means, and that will have played a part in saying whether or not statins are effective in slowing the progress of Parkinson’s.”
Momentum
With the LID-Monitor in use in four UK hospitals and the PD-Monitor playing a part in a nationwide clinical trial, the equipment seems to have got off to a strong start since its initial development three years ago, and Professor Smith hopes the momentum will continue.
“I would like to think that in the future, however it happens, the technology is in use and benefiting people living with the condition. If that’s the case then I think we’ve done our job, which is to take the research we’ve done and get it to the patients.”
The text of this article is licensed under a Creative Commons Licence. You're free to republish it, as long as you link back to this page and credit us.

Professor Stephen Smith
Research interests centred on the study of alternative representations for evolutionary algorithms and their application to problems in medicine and image processing.
Explore more research
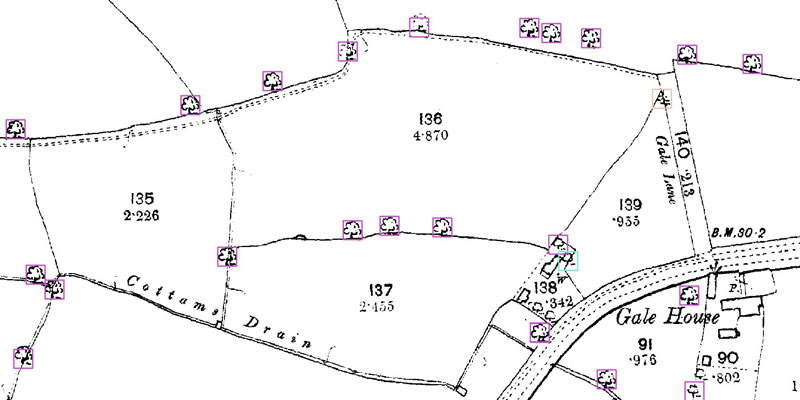
A research project needed to spot trees on historic ordnance survey maps, so colleagues in computer science found a solution.

We’re using gaming technology to ensure prospective teachers are fully prepared for their careers.

A low cost, high-accuracy device, could play a large part in the NHS's 'virtual wards'.
Research newsletter
Our monthly research newsletter features a curated mix of news, events, and recent discoveries delivered straight to your inbox.
