Understanding the mechanisms of blood cancer development
From single molecules to single cells
Lead researchers: Professor Ian Hitchcock and Dr David Kent, Department of Biology
Maintaining the correct numbers and types of mature circulating blood cells is a highly complex process. By gaining a detailed understanding of the intricate relationships between cytokine signalling, transcriptional regulation and the proliferation/differentiation of haematopoietic cells, it is possible to unravel how imbalances in this process result in disease.
Research in the groups of Professor Ian Hitchcock and Dr David Kent is focused on understanding the mechanisms of blood cell development using techniques that focus on single molecules and single stem cells.
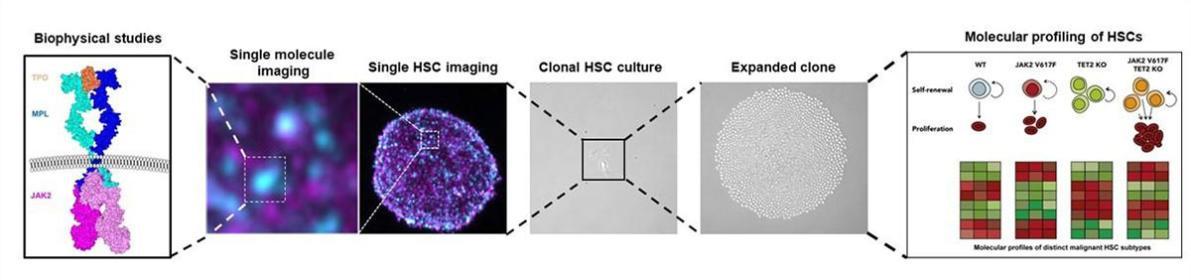
One of the simplest yet most provocative concepts in all of adult stem cell biology is how a single cell can give rise to all of the highly specialised cell types in a given tissue while retaining the capacity to make a new stem cell. At a population level, this decision making process must exist in a tightly regulated balance in order to avoid tissue degeneration (too few stem cells) or progression to cancer (too many stem cells).
Dr Kent’s lab studies how single haematopoietic stem cells (HSCs) make cell fate choices in order to understand how single cells are subverted to drive cancers and expand stem cell populations outside the body as a cell source for bone marrow transplantation and gene therapy. Current projects include determining the molecular drivers of stem cell heterogeneity, studying the physical and quantitative biology of HSCs and understanding the role of immune cell microenvironments in disease evolution and the process by which cancer evolves from a single HSC.
Many cancers are caused by mutations in genes that result in uncontrolled cell growth. The myeloproliferative neoplasms (MPNs) are a group of blood cancers characterised by the overproduction of red blood cells and/or platelets. In addition to being the most common myeloid cancer, MPNs also represent an excellent disease model to study malignant clonal expansion and mechanisms of oncogene activation, allowing the investigation of therapies that target the system-wide early dysregulation, thereby preventing transformation to more aggressive forms of leukaemia.
Research by Professor Hitchcock’s group has identified a key interaction between a cell surface receptor and a mutated protein which is essential for MPN development. Taking a novel interdisciplinary approach, the Hitchcock lab and its collaborators have determined these interactions at the level of single molecules, allowing the development of novel therapy agents to block these interactions. Other projects in Professor Hitchcock’s lab focus on understanding the causes of clotting disorders in MPN patients, bone marrow changes in non-cancerous blood diseases and how chronic infection affects platelet number and function.
References
- Lee-Six H., et al. Population dynamics of normal human blood inferred from somatic mutations. Nature. 2018 Sep;561(7724):473-478
- Shepherd MS., et al. Single-cell approaches identify the molecular network driving malignant hematopoietic stem cell self-renewal. Blood. 2018 Aug 23;132(8):791-803
- Sangkhae, V., et al. The thrombopoietin receptor, MPL, is critical for development of a JAK2V617F-induced myeloproliferative neoplasm. Blood. 2014; 124(26): 3956-3963.
- Etheridge SL., et al. JAK2V617F-positive endothelial cells contribute to clotting abnormalities in myeloproliferative neoplasms. Proc Natl Acad Sci U S A. 2014 Feb 11;111(6):2295-300
Dr David Kent and Professor Ian Hitchcock use a single cell, single molecule approach to understand the fate choices of haematopoietic stem cells and how oncogenes alter blood cell production in cancer.