Unveiling hidden pathways in photochemistry: chemical behaviour from femtoseconds to seconds
Posted on Sunday 19 April 2026
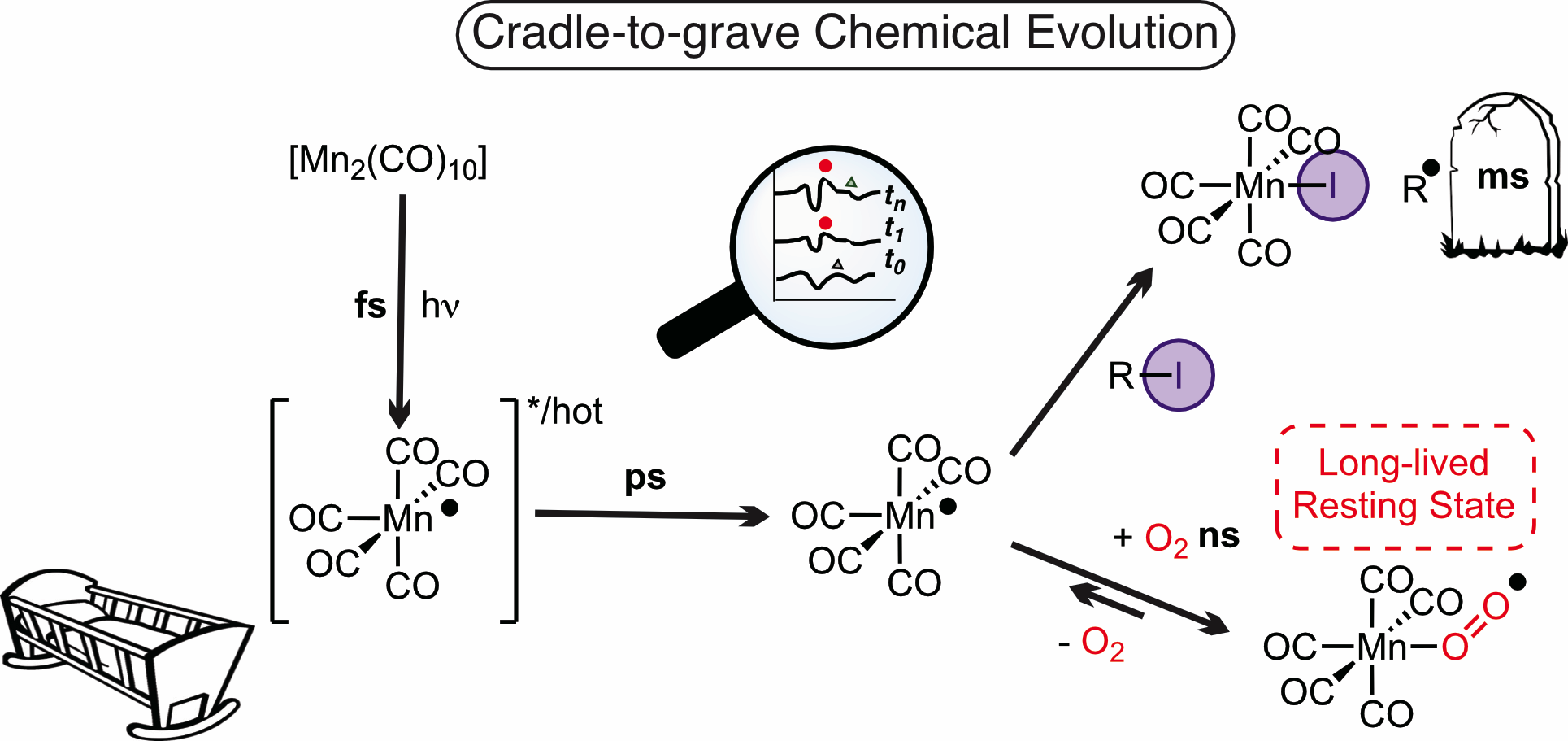
Light-driven reactions play an important role in modern synthetic chemistry, but understanding the fundamental chemical processes involved in bond activation and formation, and the exact species formed during reactions, is extremely challenging as intermediates are often very short lived. Now, a team led by Professors Ian Fairlamb and Jason Lynam at the Department of Chemistry at the University of York have developed a technique to observe hitherto unexpected species on timescales from femtoseconds to seconds.
With collaborators at the UK Central Laser facility and University College Cork (Ireland), the team used state-of-the-art laser spectroscopy to map the complete lifecycle of a manganese complex. They identified the critically important role of an oxygen-containing species which prolongs the lifetime of the active species in the reaction. As the experiments are performed under real conditions used by synthetic chemists, this cradle-to-the-grave approach provides unprecedented structural and mechanistic insights with wide-ranging consequences for the design of future, more efficient, light-driven chemical syntheses.
Notes to editors:
The paper has been published in Journal of the American Chemical Society
