News

The findings from a RSC Sustainable Laboratories grant funded project at the University of York have recently been highlighted in the Royal Society of Chemistry's Chemistry World Magazine.
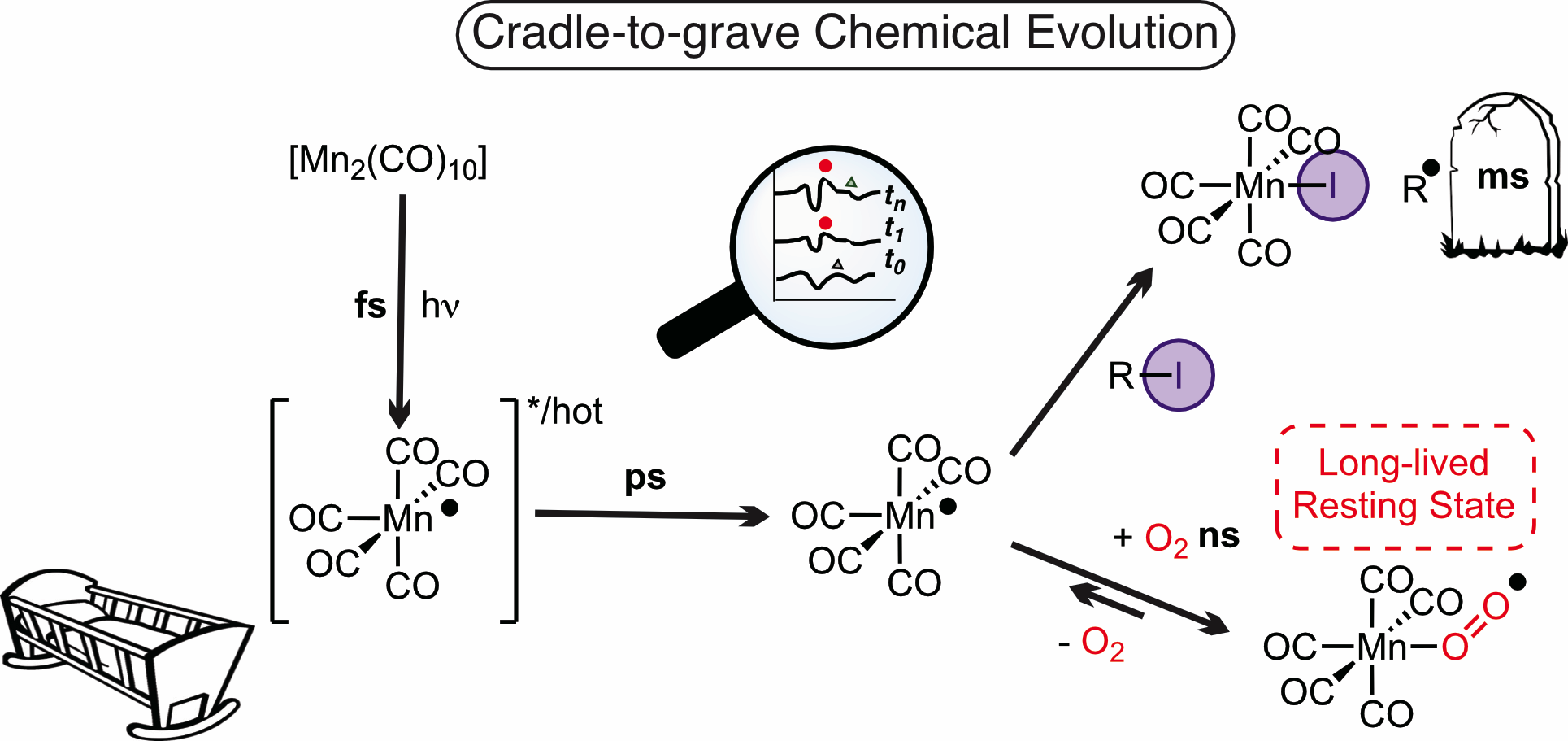
The complete lifecycle of a manganese complex as it participates in chemically important bond-activation steps has been mapped on timescales from femtoseconds to seconds. Novel, critical, and unexpected, pathways have been identified, which will inform the development of future light-activated reactions.

Often, air pollution is thought of as something that happens outside. Yet the air inside our homes may pose just as significant a risk to our health and as we typically spend 90% of our time indoors, our exposure to pollutants there is higher than outdoor exposure. This is a concern that science has, until recently, been surprisingly slow to address. The University of York is changing that, with a facility unlike anything else in the world.
Scientists say that new laboratory tests have revealed a new way to stop flu viruses, which could lead to better treatments and improved pandemic preparedness.
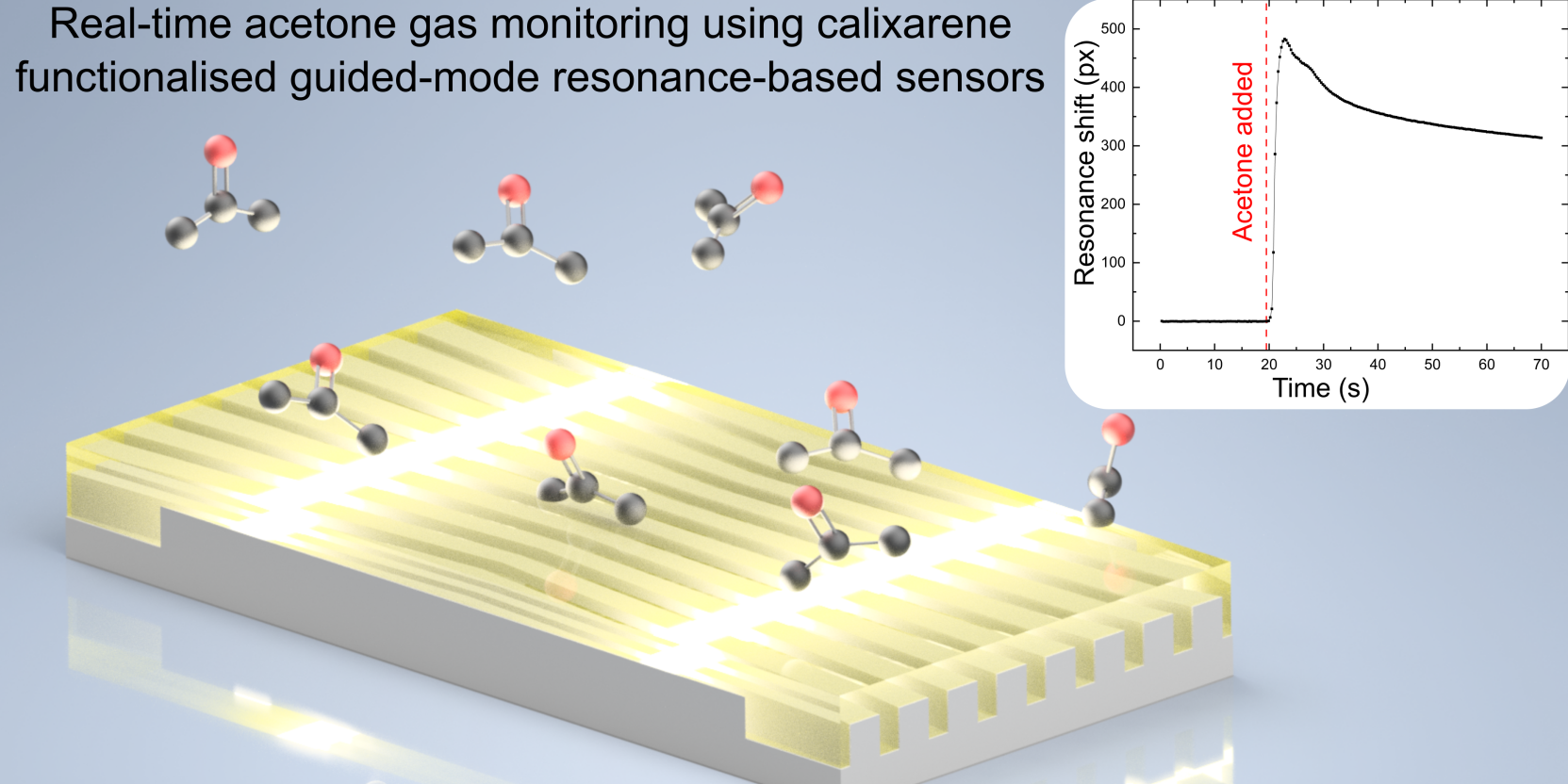
Researchers from the School of Physics, Engineering, and Technology have joined forces with the Department of Chemistry to create a new type of environmental gas sensor.
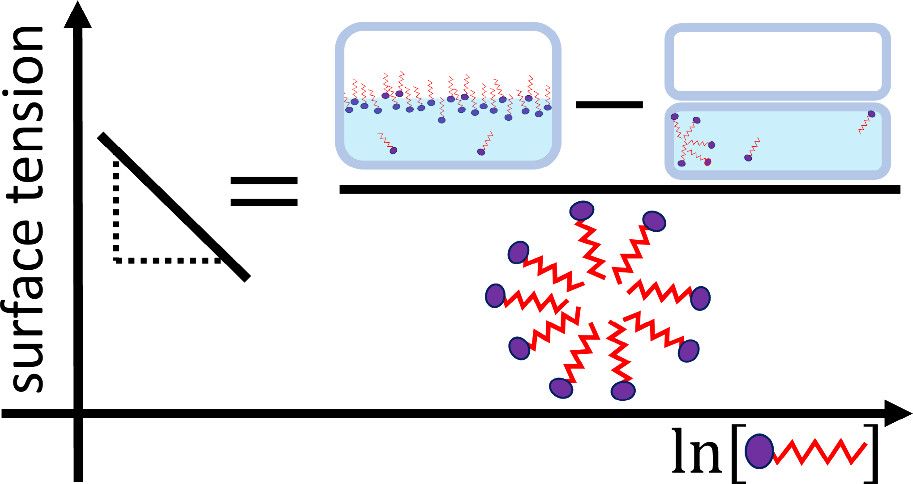
For effective cleaning we add surfactants (in soaps and detergents) to lower the surface tension of water. Using a new theory, a York chemist can explain this effect more effectively, developing new insights, and hence understand why some surfactants are much better at this job than others.

The Green Chemistry Centre has achieved and been awarded My Green Lab® Certification for Green Chemistry York at the Platinum Level.

A new record of global warming pushes our understanding back to the very start of the industrial revolution using old weather reports from sailing ships.
ChemYork Magazine
We produce a biannual magazine highlighting our teaching, research, staff and students.
Read ChemYork
