News
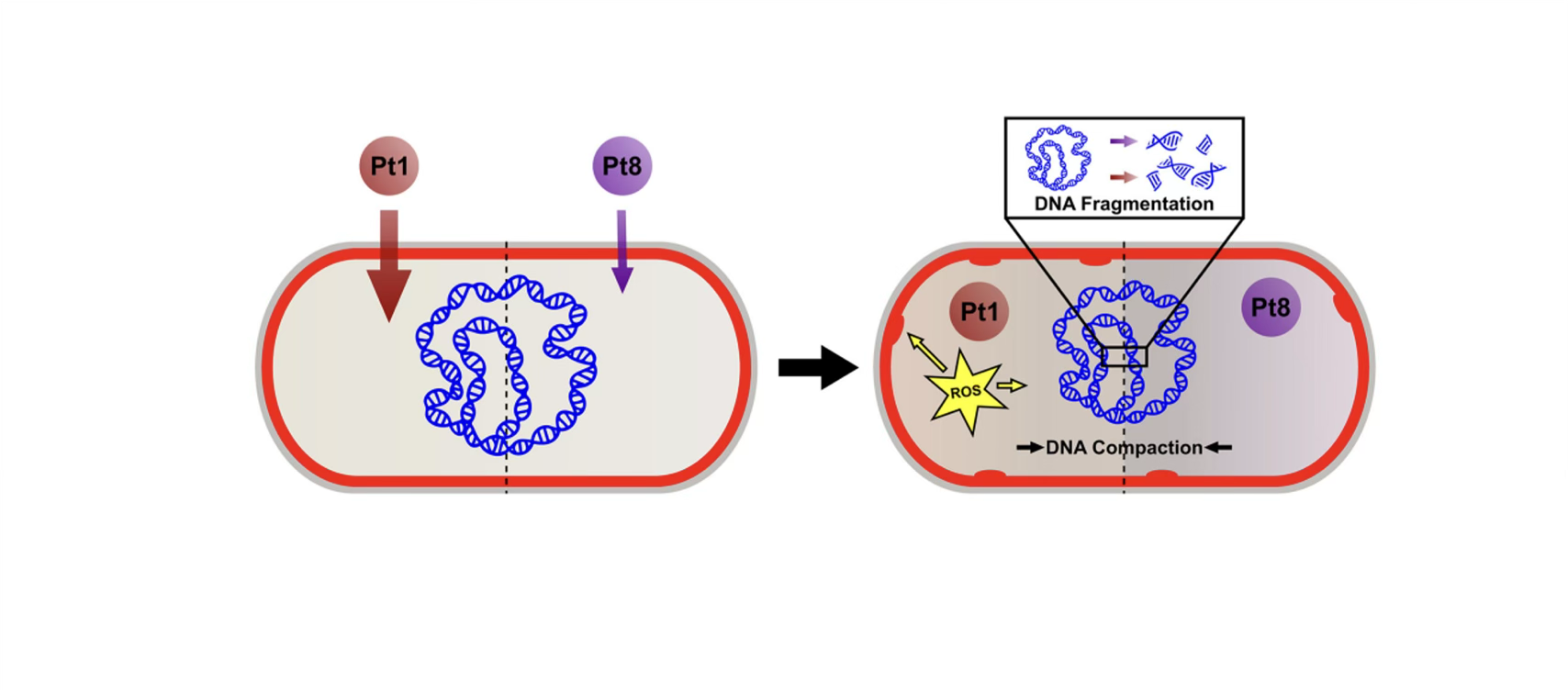
York chemists, working with international collaborators, show that small structural changes to a cisplatin-like platinum compound transform it into a non-toxic antibiotic that clears drug-resistant Staphylococcus aureus from mice skin infections.
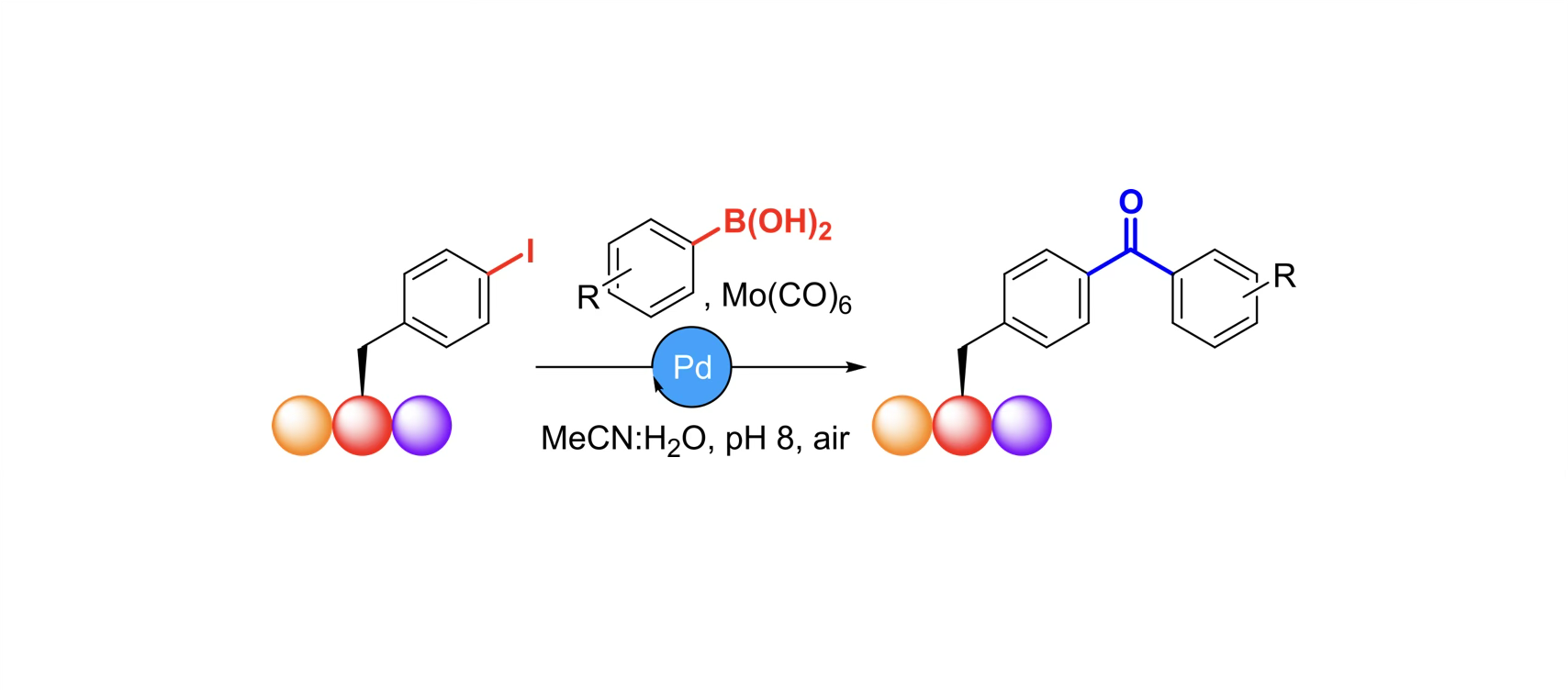
York chemists have developed a new strategy to chemically install diaryl ketones into synthetic peptides, which are at the forefront of modern drug discovery

Researchers from the University of York are part of a partnership that has been awarded more than £900,000 to transform how high-risk chemistry experiments are conducted.
Research led by the Department of Archaeology, with crucial input from the Centre of Excellence in Mass Spectrometry in the Department of Chemistry, reveals two infants buried in Roman York were laid to rest in costly purple cloth normally reserved for emperors and members of the aristocracy.
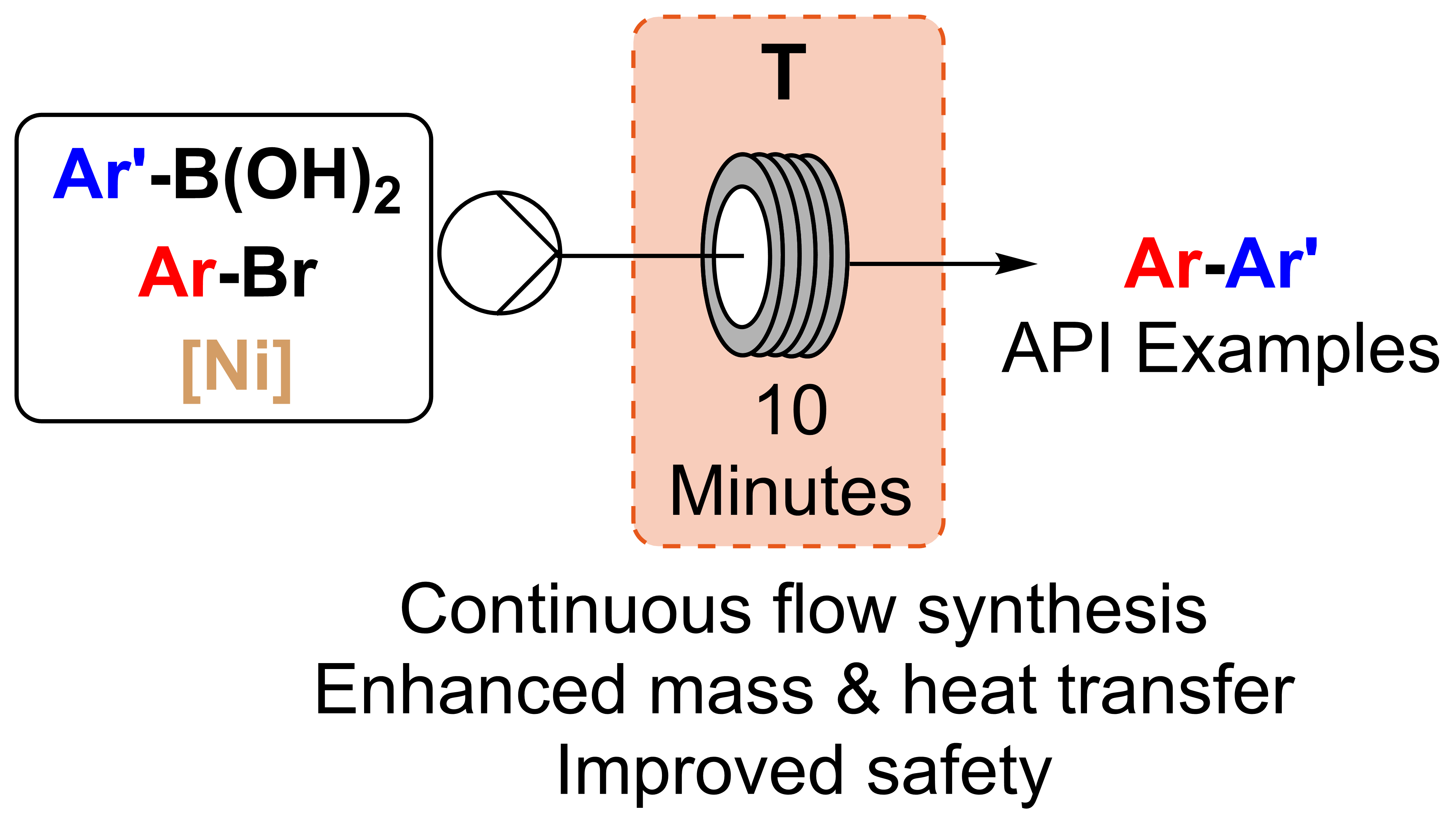
A collaboration between the University of York, the University of Leeds, and AstraZeneca has bridged chemistry and engineering to streamline drug synthesis using continuous flow technology, offering a faster, safer, and more scalable future for pharmaceutical manufacturing.

The annual poster competition for our Yr 3 PhD students has taken place, with kind sponsorship once again by Johnson Matthey. This year we were joined by Dr Paul O’Shaughnessy from JM who took time to view posters, chat with participants about their work, and give a talk on recycling platinum group metals.

The findings from a RSC Sustainable Laboratories grant funded project at the University of York have recently been highlighted in the Royal Society of Chemistry's Chemistry World Magazine.
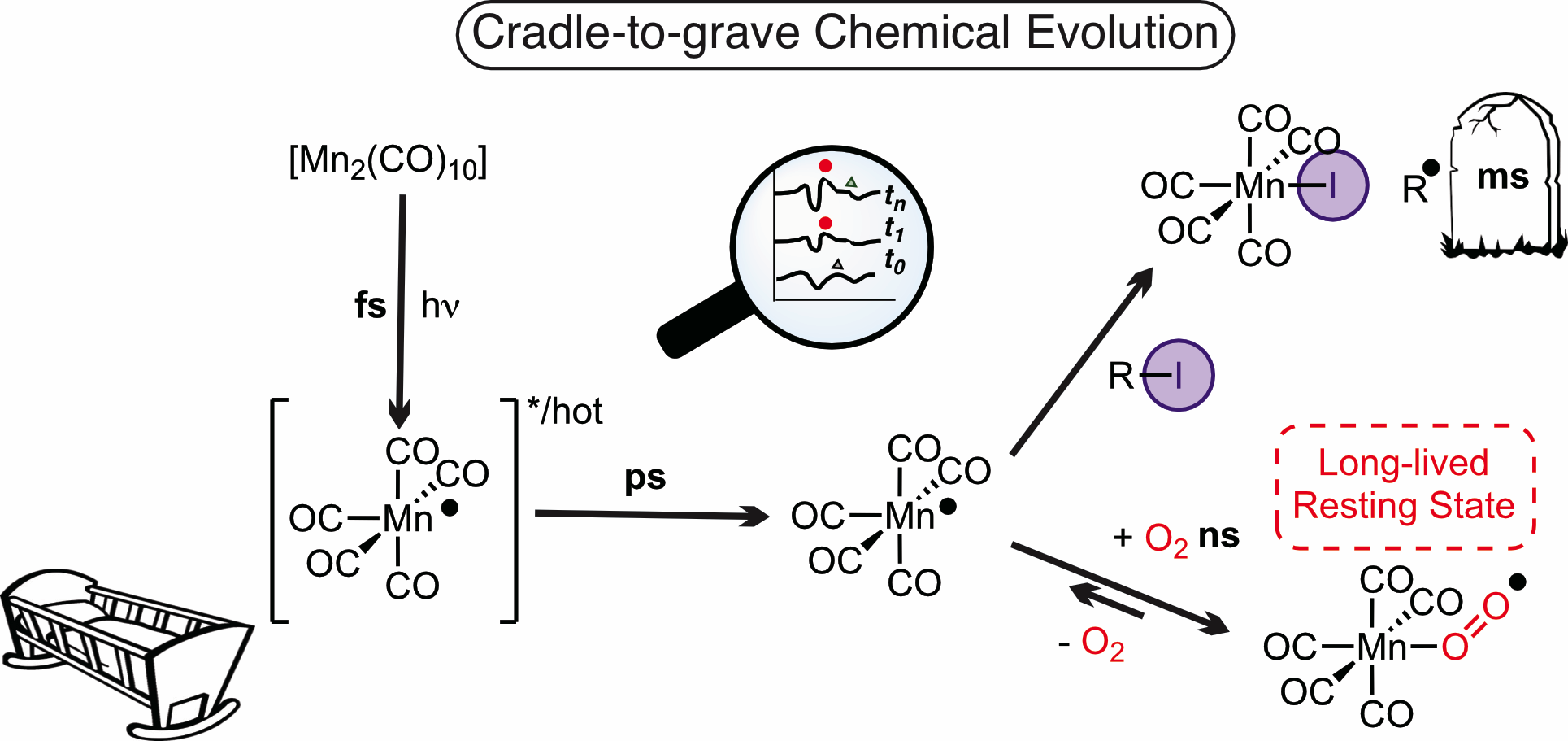
The complete lifecycle of a manganese complex as it participates in chemically important bond-activation steps has been mapped on timescales from femtoseconds to seconds. Novel, critical, and unexpected, pathways have been identified, which will inform the development of future light-activated reactions.
ChemYork Magazine
We produce a biannual magazine highlighting our teaching, research, staff and students.
Read ChemYork
