Surface Tension and Surfactants. What’s Really Going On?
Posted on Tuesday 3 March 2026
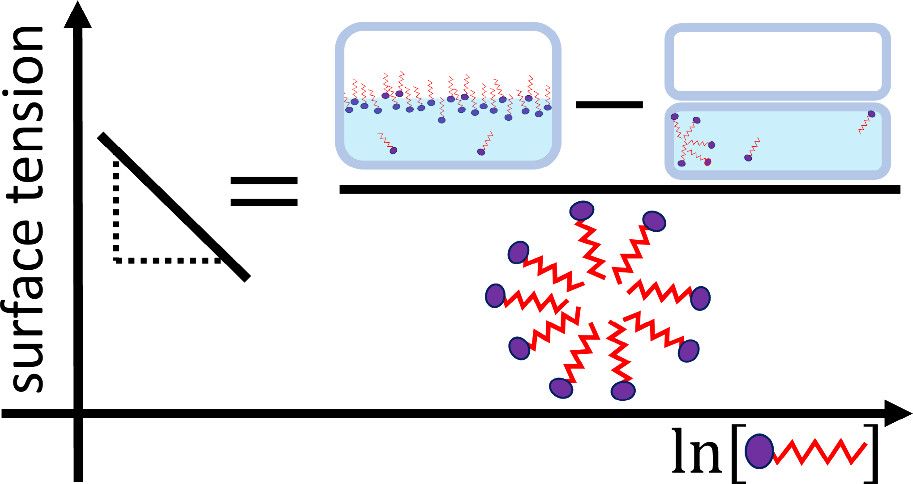
Why does surface tension decrease with increased surfactant concentration then hit a plateau? Even experts could not agree for a long time.
Seishi Shimizu (University of York) and Nobuyuki Matubayasi (Osaka University) created a new, assumption-free theory to explain the surface tension curve for all liquid solutions, including surfactants in water. They found: (1) The surface of the liquid is fully covered with surfactant molecules for much of the curve. (2) The reduction in surface tension comes from weakly-interacting surfactants that are increasing in number in the bulk. (3) Once these bulk surfactants self-assemble into micelles, their aggregation number causes the surface tension to plateau.
This new paper in Langmuir, providing new insight into such a long-established but frequently misunderstood effect, quickly became the journal’s “Most Read” article. Indeed, there are currently 3 Shimizu-Matubayasi articles in Langmuir’s ‘Top 10’, including, a novel theory for measuring surface area of materials, and a unified theory of adsorption isotherms. These papers all apply the innovative Shimizu-Matubayasi sorption theory, demonstrating exciting universality of this simple theory across wide-ranging phenomena in colloids, surfaces, materials, and liquids, and show how fundamental curiosity-led science has real-world applications.
Notes to editors:
This work has been published in Langmuir.
