Biochemistry and Biophysics
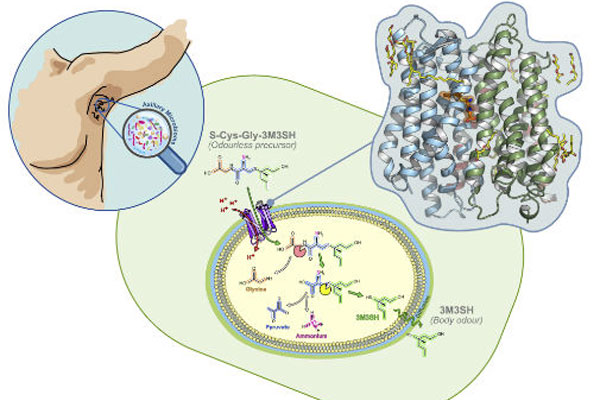
Research highlights

Decoding the opium poppy genome

Combatting land contamination with a fly gene
People
| Name | Expertise |
| Daniela Barillà |
|
| Christoph Baumann |
|
| Neil C Bruce |
|
| James Chong |
|
|
Dawn Coverley |
|
| Gareth J O Evans |
|
| Michelle Hawkins |
|
| Ian A Graham FRS |
|
| Mark Leake |
|
| Benjamin Lichman |
|
| Frans Maathius |
|
| Michael Plevin |
|
| Jennifer R Potts |
|
| Federico Sabbadin |
|
| Gavin H Thomas |
|
| Thierry Tonon |
|
| Reidun Twarock |
|
| Daniel Ungar |
|
| Robert White |
|