Dr Ines Hahn
Lecturer in Biomedical Science
Research
Research
Biography
After earning an undergraduate degree in Biology at the University of Mainz, Germany, Ines obtained her PhD in Molecular Biomedicine at the University of Bonn, Germany. She joined the University of Manchester as postdoctoral researcher and was awarded a Leverhulme Trust Early Career Fellowship in 2017. In 2022, Ines joined the Department of Biology and York Biomedical Research Institute at the University of York where she explores the coordination of cytoskeletal dynamics during neuronal development, ageing and disease. Ines is passionate about improving research culture and promoting equity, inclusion, and diversity in research, e.g. through the postdoc appreciation week initiative and as eLife Ambassador. Find her on Twitter, LinkedIN, and ORCID.
Research
Axons are the indispensable cable-like extensions of neurons that can be up to a meter long and wire our nervous system and need to be maintained for an organism's lifetime (A). Axonal development and maintenance are complex processes that are coordinated by signals from surrounding tissues (B). Those signals are then translated by signalling coordinators into controlled changes of cell behaviours. Aberration of such signalling processes can lead to neurodevelopmental and -degenerative diseases. We want to understand how signalling coordinates the cytoskeleton in axons and why misregulation causes neuronal decay.
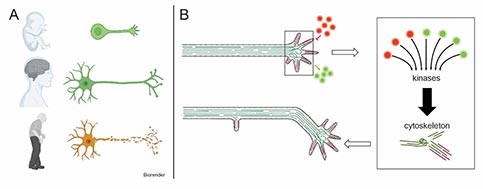
Figure 1.
Understanding the role of GSK-3β as key coordinator of neuronal development and health
GSK-3β (glycogen synthase kinase 3 β) is a key coordinator of neuronal development, because it can be triggered by wide a number of signalling cascades which it then translates into changes in neuronal cell behaviour by directly regulating the cytoskeleton (B). GSK-3β is essential for almost all steps of neuronal development and hyperactive GSK-3β is linked to neurodegenerative diseases (i.e. Alzheimer's, Parkinson's disease), schizophrenia, bipolar disorder, autism, neuropathies, and impaired regeneration after injury, making it a promising target for therapy. So far, however, therapeutic strategies manipulating GSK-3β have failed because their application was too general. We study which regulators mediate GSK-3β activity to influence neuronal development; a fundamental question which could provide the basis for refined therapies.
For this, we pioneered the use of Drosophila primary neurons for the study of GSK-3β, a powerful tool enabled us to analyse the functions of 15 microtubule (MT)- and 15 actin-binding and -regulating proteins (Hahn et al., PLoS Genet, 2021; Hahn et al., Neural dev. 2019; Qu*, Hahn* et al., eLife, 2019; Qu*, Hahn* et al. Mol Biol Cell, 2017). Using global up- or down-regulation of GSK-3β, we demonstrated that GSK-3β is a key regulator of axonal microtubules in fly and rat primary neurons. This work is funded by the Leverhulme and Wellcome Trust.
| Figure 2. Expression of active or inactive GSK-3β in primary Drosophila neurons leads to microtubule unbundling (arrow). | 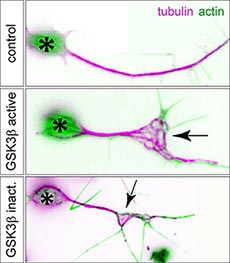 |
Understanding the impact of Neurofibromatosis type 1 on cytoskeletal regulation in neurons
Neurofibromatosis type 1 is among the most common monogenetic disorders of humans and is caused by loss of neurofibromin (Nf1), a highly conserved protein that serves as a GTPase-Activating Protein (GAP) for Ras. Loss of Nf1 leads to cognitive and behavioural deficits as well as electrophysiological abnormalities in patients and animal models. How cellular and molecular mechanisms drive these functional impairments is, however, largely unknown. We recently found that that loss of Nf1 leads to impaired neuronal outgrowth, defects in formation of pre-synapses, misregulation of the cytoskeleton in flies. We will use an interdisciplinary approach linking detailed cell mechanistic studies with functional, electrophysiological readouts; a crucial step towards identifying therapeutic interventions.
Current opportunities:
Our lab is always interested in recruiting talented and motivated scientists - please do not hesitate to get in touch: ines.hahn@york.ac.uk
Teaching and scholarship
![]()
My teaching is heavily informed by our research and I love to share my excitement for cell, developmental biology, genetics and neurobiology with students and peers. I strive to support students and researchers from diverse backgrounds to create an inclusive learning and research environment. To achieve this, I have a long track record in research culture initiatives (e.g. currently as eLife Ambassador) and outreach (e.g. through the ManFly initiative).
![]()
I enjoy interactive teaching and use current research strategies, latest outcomes and cutting-edge techniques to illustrate biological mechanisms. I have lectured in undergraduate and postgraduate programmes at the University of Manchester as well as the University of Bonn, Germany in areas of (molecular) genetics, neuroscience, cell biology and regenerative medicine.
![]()
Tutorials are an excellent way to encourage critical thinking through lively and engaged discussions in a small group setting. I aim for my tutorials to be a mix of presentations, peer-supported learning, and data analysis.
![]()
Projects in my lab are closely aligned with our research goal to understand how signalling coordinates the cytoskeleton to drive neuronal development, maintenance and disease. They will be based on our Drosophila model and include readouts ranging from detailed cell biology in primary neurons to functional/behavioural studies. With the aim to develop critical thinking and experimental skills I encourage students to develop hypotheses and design simple experiments that will to datasets resulting in a clear thesis.

Understanding how signalling coordinates the cytoskeleton is a fundamental question in cell biology with important implications for neuronal development, maintenance and disease.
