Short and long-acting insulins
Insulin derivatives that stem directly from structural work carried out within the York Structural Biology Laboratory (YSBL) are now the standard treatment for insulin-dependent diabetes for some 35 million patients worldwide. The successful development of new insulin drugs hinged upon controlling their speed of action following intravenous injection. The molecular structures of insulins were investigated using X-ray crystallography in the York Structural Biology Laboratory (YSBL) within the Department of Chemistry by the late Guy Dodson FRS, with Rod Hubbard and Jean Whittingham.
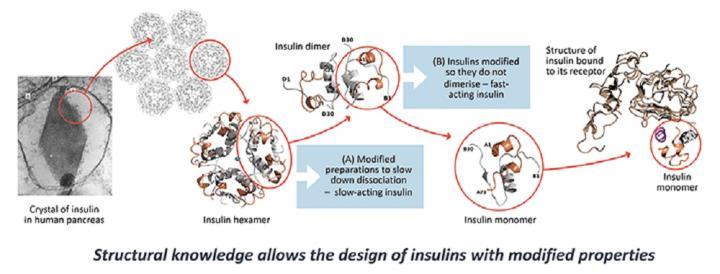
Naturally occurring insulin is stored in the pancreas in crystals which are made up of three linked pairs of insulin molecules connected to zinc ions. The response to insulin depends on how fast these aggregates break up into individual insulin molecules in the blood. From 1984 to 2000, YSBL engaged in a major collaboration on recombinant, novel modified insulins with Novo Nordisk A/S (www.novonordisk.com). The structures of these insulins provided a detailed understanding of the nature of insulin aggregation. In this way, one type of insulin was discovered that does not aggregate while another was found in which the aggregates are particularly stable. In combination, these provide the diabetic with both fast-acting and slow-acting insulin. The research has had economic impact through sales of the insulin drugs from 1999 onwards (over $6 billion in 2012) and major health impacts on diabetics worldwide. These drugs are among the top 50 drugs sold in the USA.
For further information on our research in this area please see the York Structural Biology Laboratory page.
