Dr Simon Baker
Kidney Research UK (KRUK) Fellow
Research
Much of my work involves genomic and transcriptomic methods, which are used to examine how the genes of cells can be damaged and the ways in which the genes are used in response. A particular recent focus has been the diagnostic patterns of DNA-damage left in the genome by specific events, known as “mutational signatures”. A mutational signature can be used, much like a fingerprint, to identify the causes of DNA damage and establish the relevant risk factors for different cancers.
What causes bladder cancer?
The main part of my research seeks to address this simple question. Because this question remains unanswered, there has been little progress in the prevention of bladder cancer.
The bladder epithelium (“urothelium”) spends your whole life exposed to concentrated toxins in the urine. Some of these toxins damage the DNA and we are looking at whether certain chemicals leave their signature in the DNA, in the form of specific mutation types. These mutational signatures might help explain the cause of the cancer in each individual and potentially guide treatment approaches.
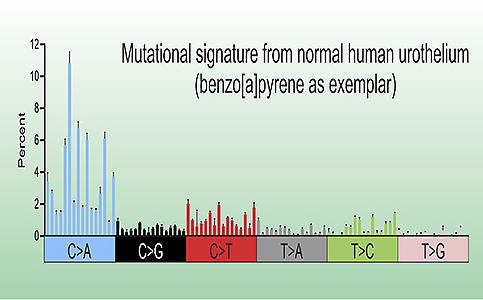
We were the first to describe a method for deriving mutational signatures from a carcinogenic exposure in an in vitro differentiated human tissue model.
Could a virus be the cause of bladder cancer?
The mutations we find in bladder tumours don’t carry the signature of chemical carcinogens. In bladder cancers the predominant mutational signatures originate from cytosine deamination by anti-viral enzymes called “APOBECs”.
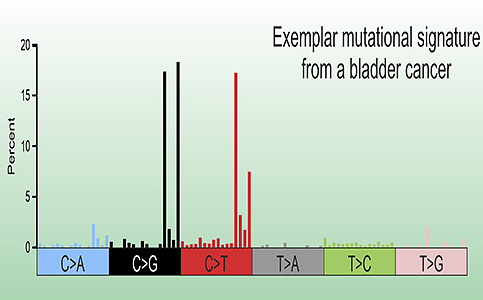
Most bladder cancer genomes harbour a mixture of the single base substitution signatures SBS2 and SBS13, which have been ascribed to APOBEC-mediated cytosine deamination.
Implicating BK polyomavirus (BKPyV)
My fellowship is looking at BK polyomavirus, which is a common childhood viral infection. BK virus is known to establish a persistent/latent infection of the kidney that survives into adulthood and can become reactivated. When BK virus becomes reactivated it can be detected in the urine. Our work has shown that BK infection of the urothelium leads to the activation of APOBECs which damage the genome, potentially leading to cancers. APOBEC3A/3B function was driven by the virus and led to damage of the host genome (quantified as apurinic/apyrimidinic sites in the genome; Oncogene 2022).
The next step is collecting the mutational signatures of viral infections and comparing them to tumours, so the risk of a BK infection can be established.
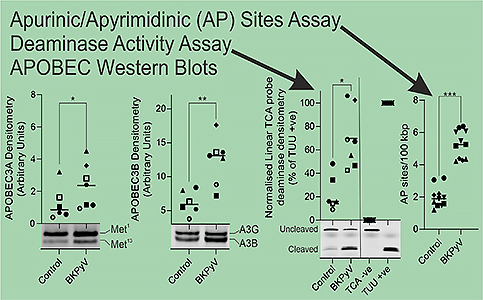
BK polyomavirus infection led to increases in APOBEC3A and APOBC3B protein, and increases in activity in deaminase functional assays. Importantly this corresponded to damage of the host urothelial genome as observed in an apurnic/apyrimidinic sites assay.
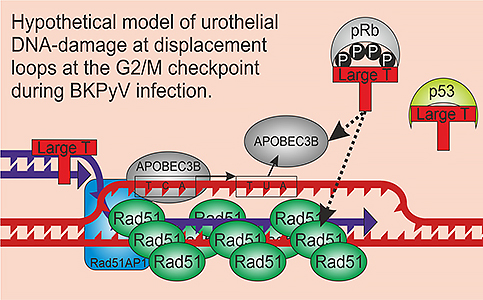
We are interested in how the APOBEC enzymes end up damaging the host genome and one hypothesis we are testing is a model where large T antigen and APOBEC3B mediate DNA damage at displacement loops formed during the G2/M arrest that occurs during BK infections. D-loops could provide an ideal single-stranded substrate for deamination.
Helping those suffering from BK infections
With our aim to improve the lives of those suffering from BK infection, our public and patient involvement (PPI) work helps develop the research. Our PPI is critical to ensure the research remains focussed and that the data drives evidence-based change in clinical practice to improve the lives of patients.
Teaching
Through my involvement with York Against Cancer, I am leading an Enthuse partnership with local schools seeking to improve cancer teaching quality and its integration throughout the secondary curriculum.
In the University I supervise undergraduate, masters and doctoral research students in their laboratory research and run tutorials.

Contact details
https://www.york.ac.uk/biology/jack-birch-unit/
