What is hyperpolarisation?
Scientists in CHyM seek to solve sensitivity problems in NMR and MRI measurements by using hyperpolarisation techniques – but what is hyperpolarisation?
Hyperpolarisation refers to a group of scientific methods which alter the magnetic properties of materials. This alteration results in large responses in nuclear magnetic resonance spectroscopy (NMR) and magnetic resonance imaging (MRI) methods. This solves the sensitivity challenges associated with NMR and MRI.
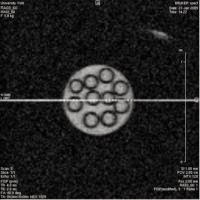
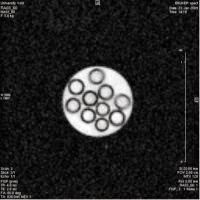
Cross section image of unhyperpolarised (left) and hyperpolarised (right) solutions containing glass tubes. The unhyperpolarised solution is generated using 128 scans, while the more intense hyperpolarised solution required only one scan.
Unhyperpolarised samples
In a normal, unpolarised sample the energy levels within a molecule created by the magnetic field of the instrument are populated almost equally. For instance, in a clinical scanner 99,999 of the molecules will be in the upper energy level while 100,000 will be in the lower energy. When the measurement is taken, effectively only 1 in 200,000 molecules are detected; the other 199,999 cancel each other out.
This distribution can be altered by either increasing the magnetic field by investing in a bigger magnet, or by hyperpolarising the sample.
Hyperpolarised samples
A hyperpolarised sample is prepared so that the proportion of molecules in each energy level are dramatically different from normal (i.e. there is a non-Boltzman distribution). This means when a measurement is performed, more of the molecules present contribute to the signal. Therefore in theory, a hyperpolarised compound in a 1.5 T clinical scanner could give a signal 200,000 times larger than normal.
The above images compare results from imaging polarised and unpolarised samples.
The first image above shows a cross section of ten glass tubes in a unpolarised liquid. This image was generated using 128 scans. The second image shows the same tubes in a hyperpolarised solution. This image was generated from a single scan. The second image appears brighter and of better quality because there is significantly more signal because the liquid is hyperpolarised.
This shows that more information can be gathered in a shorter space of time using hyperpolarised methods.
Hyperpolarisation techniques
Hyperpolarised materials can be achieved in several different ways including PHIP, SABRE, DNP and optical pumping.
Parahydrogen induced polarisation (PHIP)
In this technique parahydrogen (polarised H2 gas) can be chemically incorporated into molecules. For example alkenes can be hydrogenated to form alkanes. These alkanes will now be hyperpolarised and can be easily detected in NMR experiments. In a similar way, metal complexes which incorporate hydrogen can also be hyperpolarised in this way.
Signal Amplification by Reversible Exchange (SABRE)
A variant of PHIP, SABRE also uses parahydrogen to achieve polarised materials. A key difference of this method from PHIP is that the chemical to be studied is chemically unchanged allowing a wider variety of compounds to be studied.
Dynamic Nuclear Polarisation (DNP)
Polarisation is transferred from free radicals to chemicals. As part of this process the sample is cooled to extremely low temperatures in a strong magnetic field and exposed to microwave radiation. Once polarised, the sample is warmed to room temperature and measured in NMR or MRI measurements.
Optical pumping
Hyperpolarised gases such as helium and xenon can be prepared using laser methods.
What can hyperpolarisation be used for?
In CHyM, PHIP, SABRE and DNP are used to polarise molecules for chemical and biological applications.
General enquiries
- Centre for Hyperpolarisation in MR (CHyM)
Tel: 01904 328886
Further information
