Professor Sean T Sweeney
Professor in Neuroscience
Profile
In neurons, the endosome regulates signals controlling synapse growth. Appropriate regulation of synaptic growth is a key mechanism in refining the fidelity of synaptic communication. We work with the larval neuromuscular junction of Drosophila as a model glutamatergic synapse to identify endosomal related signaling events occurring in human pathological conditions. In parallel we use primary neurons to explore and confirm mechanisms identified in our Drosophila model
Many neurodegenerative diseases show abnormal function of endosomal compartments. My previous work has suggested that in one form of neurodegenerative disease, a childhood dementia, endosomal perturbation leads to an excessive growth of the synapse most likely due to the failure to regulate growth signals appropriately. Using this model we have identified oxidative stress as a major signal driving synapse overgrowth. We have also recently identified, with our collaborator Matthias Landgraf of the Department of Zoology, Cambridge, a critical control of synapse growth and function by the reactive oxygen species H2O2.
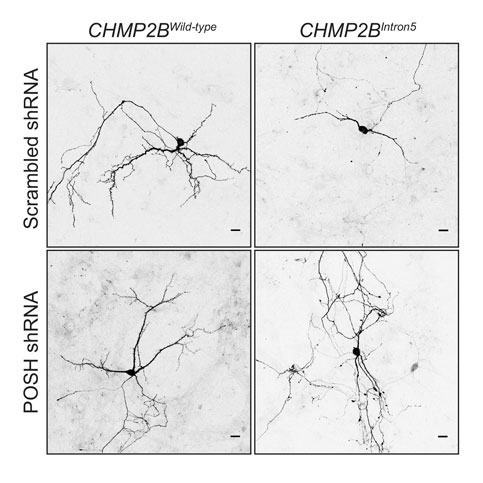
A current focus of the lab is the perturbed endosome found in Frontotemporal Dementia (FTD) and Amyotrophic Sclerosis. Both of these conditions can exist in a genetic spectrum where mutations in one gene can cause either FTD or ALS. A number of the mutant genes that cause FTD-ALS are found to encode endosomal or phagosomal proteins. We have used our Drosophila model of FTD to identify factors regulating progression of the disease and we are now building up a picture of the cellular events happening in synapses when they are affected by this condition. We have identified a pro-apoptotic signal mediated by ‘Plenty-of-SH3s’ (POSH) activated in a form of FTD-ALS. This apoptotic switch is mediated by the endosome to manipulate the early endosomal compartment in the neuron, while activating a JNK-AP1 mediated cell death.
We aim to identify events in neurons caused by mutations in FTD-ALS related genes leading to pathological progression of the condition. From this point, we can target therapeutic interventions at the ‘pathological signalling’ events that contribute to neuronal death to develop treatments for FTD-ALS.
Teaching and Scholarship
![]()
My teaching is deeply embedded in my research interests. I work to teach students from research data and techniques to give students principles of neuroscience with an experimental aspect. Bridging lecture material with experimental work refines the understanding of biological concepts and develops independent and research level thinking.
![]()
Our world is perceived through our sensory systems, taste, smell, touch, movement, sight, hearing and pain. How we detect stimuli is a refined molecular process where the physical world meets sensory neurons. In my second year lectures and practicals I explain some of the molecular processes that underpin our detection of the physical world, using our understanding invertebrate and vertebrate sensory function. In related practicals we explore the sensory function of Drosophila mutant for specific molecular sensors to determine the outcome for related behaviours of this sensory loss.
Research skills in neuroscience are taught in the third year in the integrated Masters using Drosophila to study a Parkinson's disease model based on movement. Here we develop data that is novel and at the cutting edge of research in the area to develop data handling, experimental and intellectual skills. Loss of neuronal function is associated with ageing and in my third year lectures I explain our current understanding of neurodegenerative disorders. This is where my current research interests lie, in Frontotemporal Dementia (FTD), Amyotrophic Lateral Sclerosis (ALS) and Lysosomal Storage Disease (LSD). I deliver a current understanding of these conditions up to the cutting edge of present research.
![]()
My tutorials work to explore the relationship between genetics, neuronal function and behaviour. Understanding the structure of the gene and how mutations are interpreted by the transcription and translation machinery unlocks an understanding of disease brought about by genetic change. How this genetic change relates to neurodegeneration and behaviour is drawn out in essays, writing exercises and presentations during the course of the tutorials. Writing is a key skill in conveying ideas and explaining complex principles and I aim to instil good writing practice and focus.
![]()
Projects offered are part of the 3rd year of the MBiol degree and are based on a Parkinson's disease model in Drosophila. I encourage students to develop their own projects based on a palette of tools available. These projects can be ‘blue-sky thinking’, or based on current Sweeney lab projects, but the student is encouraged to develop hypotheses, design simple experiments and develop a dataset that will result in a clear thesis that will develop critical thinking and experimental skills.



