The O'Brien group

News
December 2022
- Congratulations to Giordaina who passed her Viva!

November 2022
-
Congratulations to Ben on the successful defense of his thesis! We wish Ben well as he embarks on a his career at Apex Molecular.

September 2022
- Congratulations to Giordaina for winning yet another prize - the Astex Pharmaceuticals poster competition at Astex, Cambridge!
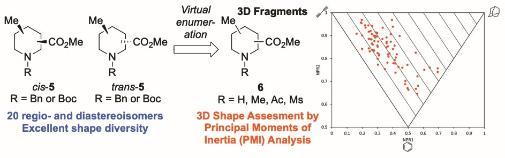
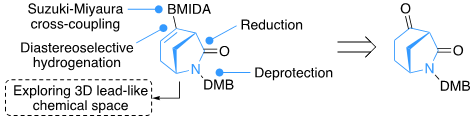
- Our latest paper on the "Exploration of piperidine 3D fragment chemical space: synthesis and 3D shape analysis of fragments derived from 20 regio- and diastereoisomers of methyl substituted pipecolinates" been published in RSC Medicinal Chemistry.
July 2022
- Congratulations to Giordaina for winning a Roger J Mawby Demonstrator Award, for the best deomstrators in the undergraduate teaching labs!

- Giordaina wins again - this time she delivered the best organic chemistry talk at the Sygnature Discovery Postgraduate Symposium. Congratulations!

June 2022
- Congratulations to Stephen on delivering an excellent lecture at the organic plenary session
- Well done to Jake and Will on completing their MChem projects - celebrated with a group lunch.

May 2022
- Our latest paper on Escape from planarity in fragment-based drug discovery: A synthetic strategy analysis of synthetic 3D fragment libraries has been published in Drug Discovery Today. Congratualations Hanna!
April 2022
-
Peter delivered his first talk since lockdown for the Musgrave Lecture Symposium at Durham.
- Welcome to Giordaina who starts as a postdoc in the group, working on inhibitors of the Nsp3 macrodomain of SARS-CoV-2.
March 2022
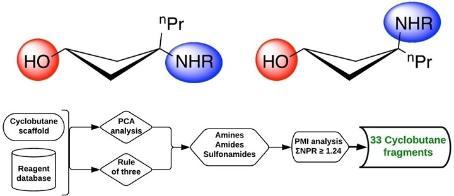
- The groups latest paper, on 'Puckering the Planar Landscape of Fragments: Design and Synthesis of a 3D Cyclobutane Fragment Library' has been published in collaboration with Dr. Maikel Wijtmans from the Amsterdam Institute of Molecular and Life Sciences (AIMMS). Congratulations to Hanna.
February 2022
- Welcome to Ben who starts as a postdoc in the group, working on the development of air stable organometallic gels in collaberation with Prof. Dave Smith.
- The groups latest paper is available as a preprint 'Organolithium Gels – Simple Easily Divided Delivery Vehicles for Highly Reactive Species'. This work is the beginning of a collaboration with Prof. Dave Smith.
December 2021
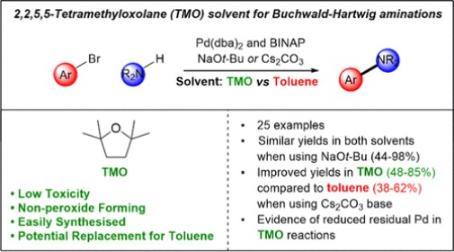
- The groups latest paper, on the use of '2,2,5,5-Tetramethyloxolane (TMO) as a Solvent for Buchwald–Hartwig Aminations' has been published in collaboration with Dr Tom Farmer. Congratulations to Ben.
September 2021
- Welcome to our new PhD students, Lucy, Stuart M and Stuart S (joint with Prof. Ian Fairlamb). They will be working on the sp2-sp3 Suzuki-Miyaura coupling of heterocyclic aliphatic boronates.
- Welcome to new MRes student Hannah, who will working on Novel 3-D Building Blocks for Elaboration of Fragments to Lead-like Compounds.
June 2021
- The groups latest paper, on the use of 'Escape from planarity in fragment-based drug discovery: A physicochemical and 3D property analysis of synthetic 3D fragment libraries' has been published in collaboration with Iwan J.P. de Esch. Congratulations to Hanna.
April 2021
-
Peter delivered a department seminar on his Royal Society Industry Fellowship. Here, he was the warm up act for the invited external speaker, Dr Susannah Coote, a York alumnus who carried out her PhD in the group and is now a lecturer at Lancaster University.
- The group’s latest covid-19 related work was published in Science Advances – we had four York 3D fragment hits against the macrodomain protein.
March 2021
-
Stephen Yao and Giordaina Hartley presented posters on their PhD projects at the Catalysis@York event organised by Andy Weller. It was a great afternoon of science showcasing the wide-ranging catalysis research in the Department
-
Peter gave an invited lecture at the RSC’s meeting entitled “Synthesis in Drug Discovery and Development” on the topic of his Royal Society Industry Fellowship
February 2021
-
Congratulations to Hannah on the successful completion of her MSc on sulfoximine chemistry - not even the coronavirus pandemic could stop her!
January 2021
-
The group’s latest paper on “Synthesis and functionalisation of a bifunctional normorphan 3D building block for medicinal chemistry” is published in Tetrahedron. Congratulations to Andres, James D and James F.
December 2020
-
Peter gave a virtual research lecture to over 100 chemists at Pfizer in the US and UK.
-
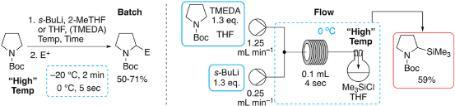
The group’s latest lithiation paper on “Rapid “high” temperature batch and flow lithiation-trapping of N-Boc pyrrolidine” is published in Tetrahedron. Congratulations to Alice and James.
November 2020
-
Giordaina represented the group and presented her sulfoximine results at the virtual RSC Organic Division Poster Competition
-
Congratulations to Hanna on the successful defense of her thesis, with Professor Taylor carrying out possibly his last ever PhD viva! We wish Hanna well as she embarks on a post-doctoral position in the group of Professor Fredrik Almqvist at Umea in Sweden.
-
We welcome PhD student Andres (ex MSc student!) to the group for a later-than-planned start to his PhD studies.
October 2020
-
We welcome MChem project students Lucy and Rachel to the group.
-
The group’s paper on “Crystallographic and electrophilic fragment screening of the SARS-CoV-2 main protease” is published in Nature Commun. Congratulations to Tom, Paul, Hanna and James.
September 2020
-
Thank you and goodbye to post-doc Kevin who leaves York after over three and half years to return to St Andrews to a post-doc position with Professor Andrew Smith.
-
Congratulations to Sophie on the successful defense of her electrochemical thesis, co-supervised by Victor Chechik and Alison Parkin. Sophie left the group earlier in the year to take up an industrial position at Apex Molecular. We wish her the best of luck in her new job. Here’s a photo of some of us on the celebratory post-viva Zoom call.
August 2020
-
Peter gave a virtual research lecture at a Future Technologies for Covid-19 Eventhosted by the University of York. He presented the group’s fragment-based drug discovery work including his Royal Society Industry Fellowship and the exciting recent fragment hits against Covid-19 proteins.
May 2020
-
Congratulations to Nico on the successful defense of his organolithium thesis. Nico left the group to do a post-doc with Dr Chris Spicer in York before moving on to an industrial position at Concept Life Sciences. Good luck to Nico in the new job!
-
The group’s work on 3-D fragments and their use in potential drug discovery starting points for treating covid-19 featured in a University news story.
April 2020
-
The group’s paper on “Design and Synthesis of 56 Shape Diverse 3-D Fragments” is published in Chem. Eur. J. Congratulations to the 3-D fragments team for a several-year odyssey.
January 2020
-
Congratulations to Kevin and Nico on their excellent organolithium review that features in the 100th volume of Organic Reactions, alongside an illustrious list of other authors in the same volume.

News archive
Peter O'Brien
Peter O'Brien's Biography
Peter O'Brien studied for a degree and PhD at the University of Cambridge, carrying out a PhD under the supervision of Stuart Warren. After the award of his PhD in 1995, he moved to the University of York as a Royal Commission for the Exhibition of 1851 Research Fellow. In March 1996, he was appointed as a lecturer at York and was promoted to Senior Lecturer (2002), Reader (2005) and Professor (2007).
His research interests include asymmetric synthesis, organolithium methodology, synthesis of saturated heterocycles, medicinal chemistry and fragment-based drug discovery – and his efforts in these areas have been recognized by the award of the Royal Society of Chemistry Organic Stereochemistry Award in 2013 and the AstraZeneca, GlaxoSmithKline, Pfizer & Syngenta prize for Process Chemistry Research in 2017. In 2019, he has awarded a Royal Society Industry Fellowship to work in collaboration with AstraZeneca on fragment-based drug discovery in 3-dimensions.
Peter is also a passionate teacher and was awarded a Vice-Chancellor’s Teaching Award in 2015 and the University’s Teacher of the Year in the York University Student Union Excellence Awards in 2019.
Additional information:
Earlier in his career, Peter was awarded one of the Royal Society of Chemistry's Meldola medals and prizes (1999) and a GlaxoWellcome award for innovative organic chemistry (2000).
Peter has also been an active member of the Royal Society of Chemistry's Heterocyclic & Synthesis Group, serving as the group’s Secretary in 2005-2007 and the Chairperson in 2017-2020.
Peter was an associate editor of the international journal “Tetrahedron” from 1998-2017.

Peter presenting at the Royal Society to launch his industry fellowship with AstraZeneca, November 2019
Publications
Publication List (1995 - Present)
S. P. Jones, J. D. Firth, M C. Wheldon, M. Atobe,a R. E. Hubbard, D. C. Blakemore C. De Fusco, S. C. C. Lucas, S. D. Roughley, L. R. Vidler, M. A. Whatton, A. J.-A. Woolford, G. L. Wrigley and P. O'Brien. RSC Med. Chem., 2022, 27, 2484

H. F. Klein, D. J. Hamilton, I. J. P. de Esch, M. Wijtmans, P. O'Brien. Drug Discovery Today, 2022, DOI: 10.1016/j.drudis.2022.05.021
D. J. Hamilton, M. Beemsterboer, C.e M. Carter, J. Elsayed, R. E. M. Huiberts, H. F. Klein, P. O'Brien, I. J. P. de Esch, M. Wijtmans. ChemMedChem, 2022, e2022001
150 "Organolithium Gels – Simple Easily Divided Delivery Vehicles for Highly Reactive Species"
P. Slavik, P. O'Brien, D. K. Smith, ChemRxiv, 2022, 10.26434/chemrxiv-2022-q2txr
149 "2,2,5,5-Tetramethyloxolane (TMO) as a Solvent for Buchwald–Hartwig Aminations"
B. R. Trowse, F. P. Byrne, J. Sherwood, P. O’Brien, J. Murray, and T. J. Farmer, ACS Sustainable Chem. Eng. 2021, 9, 17330

D. J.Hamilton, T. Dekker, H. F. Klein, G. V. Janssen, M. Wijtmans, P. O’Brien, I. J.P.de Esch, Drug Discov. Today Technol. 2020, 38, 77
M. Schuller, G. J. Correy, S. Gahbauer, D. Fearon, T. Wu, R. Efraín Díaz, I. D. Young, L. Carvalho Martins, D. H. Smith, U. Schulze-Gahmen, T. W. Owens, I. Deshpande, G. E. Merz, A. C. Thwin, J. T. Biel, J. K. Peters, M. Moritz, N. Herrera, H. T. Kratochvil, QCRG Structural Biology Consortium, A. Aimon, J. M. Bennett, J. Brandao Neto, A. E. Cohen, A. Dias, A. Douangamath, L. Dunnett, O. Fedorov, M. P. Ferla, M. R. Fuchs, T. J. Gorrie-Stone, J. M. Holton, M. G. Johnson, T. Krojer, G. Meigs, A. J. Powell, J. Gregor, M. Rack, V. L. Rangel, S. Russi, R. E. Skyner, C. A. Smith, A. S. Soares, J. L. Wierman, K. Zhu, P.O’brien, N. Jura, A. Ashworth, J. J. Irwin, M. C. Thompson, J. E. Gestwicki, F. Von Delft, B. K. Shoichet, J.s S. Fraser, I. Ahel, Sci. Adv. 2021, 7, eabf8711
A. R. Gomez-Angel, J. R. Donald, J. D. Firth, C. De Fusco, R. I. Storer, D. J. Cox and P. O’Brien, Tetrahedron, 2021, 81, 131961.

145 “Rapid “high” temperature batch and flow lithiation-trapping of N-Boc pyrrolidine”
A. Kwong, J. D. Firth, T. J. Farmer and P. O’Brien, Tetrahedron, 2021, 81, 131899.

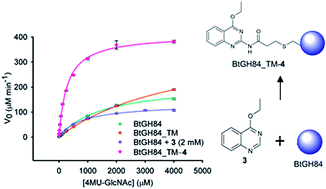
144 “Fragment-derived modulators of an industrial β-glucosidase”
E. Makraki, J. F. Darby, M. G. Carneiro, J. D. Firth, A. Heyam, E. Ab, P. O’Brien, G. Siegal and R. E. Hubbard, Biochem. J. 2020, 477, 4383-4395.
143. “Crystallographic and electrophilic fragment screening of the SARS-CoV-2 main protease”
A. Douangamath, D. Fearon, P. Gehrtz, T. Krojer, P. Lukacik, C. D. Owen, E. Resnick, C. Strain-Damerell, A. Aimon, P. Ábrányi-Balogh, J. Brandaõ-Neto, A. Carbery, G. Davison, A. Dias, T. D Downes, L. Dunning, M. Fairhead, J. D. Firth, S. P. Jones, A. Keely, G. M. Keserü, H. F Klein, M. P. Martin, M. E. M. Noble, P. O’Brien, A. Powell, R. Reddi, R. Skyner, M. Snee, M. J. Waring, C. Wild, N. London, F. von Delft and M. A. Walsh, Nat. Commun. 2020, 11, 5047.
142. “Design and Synthesis of 56 Shape Diverse 3-D Fragments”
P. O’Brien, T. D. Downes, S. P. Jones, H. F. Klein, M. C. Wheldon, M. Atobe, P. S. Bond, J. D. Firth, N. S. Chan, L. Waddelove, R. E. Hubbard, D. C. Blakemore, C. De Fusco, S. D. Roughley, L. R. Vidler, M. A. Whatton, A. J.-A. Woolford and G. L. Wrigley, Chem. Eur. J., 2020, 26, 8969-8975.
H. E. Ho, A. Pagano, J. A. Rossi-Ashton, J. R. Donald, R. G. Epton, J. C. Churchill, M. J. James, P. O’Brien , R. J. K. Taylor and W. P. Unsworth, Chem. Sci. 2020, 11, 1353-1360.
140. “Enantioselective Lithiation–substitution of nitrogen-containing heterocycles”
K. Kasten, N. Seling and P. O’Brien, Org. React. 2019, 100, 255-328.
H. E. Ho, T. C. Stephens , T. J. Payne, P. O’Brien , R. J. K. Taylor and W. P. Unsworth. ACS Catal., 2019, 9, 504-510
J. D. Firth and P. O'Brien. Chemical and Biological Synthesis: Enabling Approaches for Understanding Biology, 2018, pp. 74-113 DOI: 10.1039/9781788012805-00074

J. D. Firth, G. Gelardi, P. J. Rayner, D. Stead and P. O'Brien, Heterocycles, 2018, 97, 1288-1303.

H. E. Ho, M. J. James, P. O’Brien, R. J. K. Taylor and W. P. Unsworth, Org. Lett. 2018, 20, 1439–1443.
135. "Gram-Scale Synthesis of the (−)-Sparteine Surrogate and (−)-Sparteine"
J. D. Firth, S. J. Canipa, L. Ferris and P. O'Brien. Angew. Chem. Int. Ed. 2018, 57, 223-226.
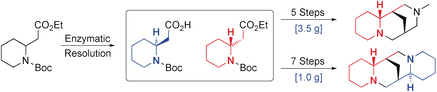
134. "Increase of enzyme activity through specific covalent modification with fragments"
J. F. Darby, M. Atobe, J. D. Firth, P. Bond, G. J. Davies, P. O'Brien and R. E. Hubbard, Chem. Sci., 2017, 8, 7772-7779
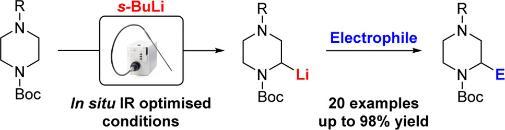
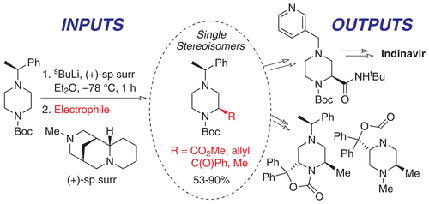
133 "General Procedures for the Lithiation/Trapping of N-Boc Piperazines"
J. D. Firth, P. O’Brien and L. Ferris, J. Org. Chem., 2017, 82, 7023–7031
M. J. James, N. D. Grant, P. O’Brien, R. J. K. Taylor and W. P. Unsworth, Org. Lett. 2016, 18, 6256-6259.
A. K. Clarke, M. J. James, P. O'Brien, R. J. K. Taylor and W. P. Unsworth, Angew. Chem. Int. Ed. 2016, 55, 13798-13802.
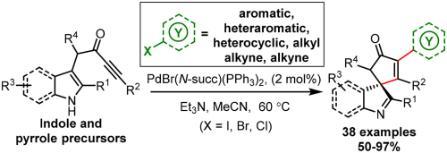
130 "Selective Synthesis of Six Products from a Single Indolyl α-Diazocarbonyl Precursor"
M. J. James, P. O'Brien, R. J. K. Taylor and W. P. Unsworth, Angew. Chem. Int. Ed. 2016, 55, 9671-9675.
J. T. R. Liddon, M. J. James, A. K. Clarke, P. O'Brien, R. J. K. Taylor and W. P. Unsworth, Chem. Eur. J. 2016, 22, 8777-8780.
S. J. Chambers, G. Coulthard, W. P. Unsworth, P. O'Brien and R. J. K. Taylor, Chem. Eur. J, 2016, 22, 6496-6500
J. D. Firth, P. O’Brien and L. Ferris, J. Am. Chem. Soc., 2016, 138, 651-659.
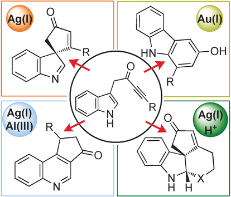
126 “Synthesis of Spirocyclic Indolenines”
M. J. James, P. O’Brien, R. J. K. Taylor and W. P. Unsworth, Chem. Eur. J. 2016, 22, 2856-2881.
P. J. Rayner, J. C. Smith, C. Denneval, P. O’Brien, P. A. Clarke and R. A. J. Horan, Chem. Commun. 2016, 52, 1354-1357.
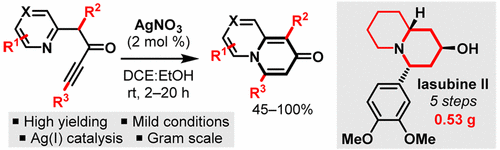
124 “Silver(I)-Catalysed Dearomatisation of Alkyne-Tethered Indoles: Divergent Synthesis of Spirocyclic Indolenines and Carbazoles”
M. J. James, R. E. Clubley, K. Y. Palate, T. J. Procter, A. C. Wyton, P. O’Brien, R. J. K. Taylor and W. P. Unsworth, Org. Lett., 2015, 17, 4372-4375.
123 “Investigation of the Lithiation-trapping of a N-Boc Bispidine-ketal: Reactivity and Diastereoselectivity”
D. Stead, P. O’Brien and A. Sanderson, Synlett, 2015, 26, 2381-2384.
122 “Silver(I) or copper(II)-mediated dearomatisation of aromatic ynones: direct access to spirocyclic scaffolds”
M. J. James, J. D. Cuthbertson, P. O’Brien, R. J. K. Taylor and W. P. Unsworth, Angew. Chem. Int. Ed., 2015, 54, 7640-7643.
121 “Lead-oriented synthesis: investigation of organolithium-mediated routes to 3-D scaffolds and 3-D shape analysis of a virtual lead-like library”
M. Lüthy, M. C. Wheldon, C. Haji-Cheteh, M. Atobe, P. S. Bond, P. O’Brien,R. E. Hubbard and I. J. S. Fairlamb, Bioorg. Med. Chem., 2015, 23, 2680-2694.
120 “Cross-Coupling Knows No Limits: Assessing the Synthetic Potential of the Palladium-Catalysed Cross-Coupling of Organolithiums”
J. D. Firth and P. O’Brien, ChemCatChem, 2015, 7, 395-397.
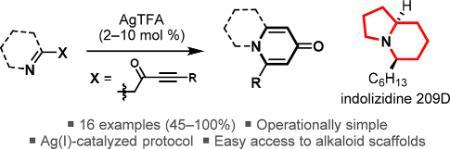
119 “Revisiting the sparteine surrogate: development of a resolution route to the (–)-sparteine surrogate”
J. D. Firth, P. O’Brien and L. Ferris, Org. Biomol. Chem. 2014, 12, 9357-9365.
118 “Use of copper(II)/diamine catalysts in the desymmetrisation of meso-diols and asymmetric Henry reactions: comparison of (–)-sparteine and (+)-sparteine surrogates”
S. J. Canipa, A. Stute and P. O’Brien, Tetrahedron, 2014, 70, 7395-7403.
117 “On the synthesis of a-amino sulfoxides”
P. J. Rayner, G. Gelardi, P. O’Brien, R. A. J. Horan and D. C. Blakemore, Org. Biomol. Chem. 2014, 12, 3499-3512.
116 “Asymmetric Lithiation-Trapping of N-Boc Heterocycles at Temperatures above –78 °C”
G. Gelardi, G. Barker, P. O’Brien and D. C. Blakemore, Org. Lett. 2013, 15, 5424-5427.
115 “Preparation and Reactions of Enantiomerically Pure a-Functionalised Grignard Reagents”
P. J. Rayner, P. O’Brien and R. J. Horan, J. Am. Chem. Soc. 2013, 135, 8071-8077.
114 “Halogen-Bonded Liquid Crystals of 4-Alkoxystilbazoles with Molecular Iodine: a Very Short Halogen Bond and Unusual Mesophase Stability”
L. J. McAllister, C. Präsang, J. P.-W. Wong, R. J. Thatcher, A. C. Whitwood, B. Donnio, P. O'Brien, P. B. Karadakov and D. W. Bruce, Chem. Commun. 2013, 49, 3946-3948.
113 “Stereocontrolled Synthesis of the AB Rings of Samaderine C”
D. J. Burns, S. Mommer, P. O’Brien, R. J. K. Taylor, A. C. Whitwood, and S. Hachisu, Org. Lett., 2013, 15, 394-397.
112 “Synthesis of P-Stereogenic Phospholene Boranes via Asymmetric Deprotonation and Ring-Closing Metathesis”
X. Wu, P. O’Brien, S. Ellwood, F. Secci and B. Kelly, Org. Lett., 2013, 15, 192-195.
111 “Amines bearing tertiary substituents by tandem enantioselective carbolithiation–rearrangement of vinyl ureas”
M. Tait, M. Donnard, A. Minassi, J. Lefranc, B. Bechi, G. Carbone, P. O’Brien and J. Clayden, Org. Lett., 2013, 15, 34-37.
110 “Catalytic asymmetric synthesis of butane diacetal-protected (4S,5S)-dihydroxycyclohexen-1-one and use in natural product synthesis”
D. J. Burns, S. Hachisu, P. O’Brien and R. J. K. Taylor, Org. Biomol. Chem., 2012, 10, 7666-7668.
109 “An Experimental and In Situ IR Spectroscopic Study of the Lithiation–Substitution of N-Boc 2-phenylpyrrolidine and piperidine: Controlling the Formation of Quaternary Stereocenters”
N. S. Sheikh, D. Leonori, G. Barker, J. D. Firth, K. R. Campos, P. O’Brien and I. Coldham, J. Am. Chem. Soc., 2012, 134, 5300-5308.
108 “Enantioselective, Palladium-Catalyzed a-Arylation of N-Boc Pyrrolidine: In Situ React IR Spectroscopic Monitoring, Scope and Synthetic Applications”
G. Barker, J. L. McGrath, A. Klapars, D. Stead, G. Zhou, K. R. Campos and P. O’Brien, J. Org. Chem. 2011, 76, 5936-5953.
107 “One-ligand Catalytic Asymmetric Deprotonation of a Phosphine Borane: Synthesis of P-Stereogenic Bisphosphine Ligands”
J. Granander, F. Secci, S. J. Canipa, P. O’Brien and B. Kelly, J. Org. Chem. 2011, 76, 4794-4799.
106 “Synthesis of the Azaphilones (+)-Sclerotiorin and (+)-8-OMethylsclerotiorinamine Utilizing (+)-Sparteine Surrogates in Copper-Mediated Oxidative Dearomatization”
A. R. Germain, D. M. Bruggemeyer, J. Zhu, C. Genet, P. O’Brien and J. A. Porco, Jr., J. Org. Chem. 2011, 76, 2577-2584.
105 “Investigation of bispidines as the stoichiometric ligand in the two-ligand catalytic asymmetric deprotonation of N-Boc pyrrolidine”
G. Barker, P. O’Brien and K. R. Campos, ARKIVOC 2011, (v), 217-229.
104 “Asymmetric Deprotonation using s-BuLi or i-PrLi and Chiral Diamines in THF: the Diamine Matters”
G. Carbone, P. O’Brien and G. Hilmersson, J. Am. Chem. Soc., 2010, 132, 15445-15450.
103 “Synthesis of P-Stereogenic Compounds via Kinetic Deprotonation and Dynamic Thermodynamic Resolution of Phosphine Sulfides: Opposite Sense of Induction using (–)-Sparteine”
J. J. Gammon, V. H. Gessner, G. R. Barker, J. Granander, A. C. Whitwood, C. Strohmann, P. O’Brien and B. Kelly, J. Am. Chem. Soc., 2010, 132, 13922-13927.
102 “Diamine-free Lithiation-Trapping of N-Boc Heterocycles using s-BuLi in THF”
G. Barker, P. O’Brien and K. R. Campos, Org. Lett. 2010, 12, 4176-4179.
101 “Asymmetric Synthesis via Aziridinium Ions: Exploring the Stereospecificity of the Ring Opening of Aziridinium Ions and a Formal Synthesis of (–)-Swainsonine”
S. J. Oxenford, S. P. Moore, G. Carbone, G. Barker, P. O’Brien, M. R. Shipton, J. Gilday and K. R. Campos, Tetrahedron: Asymmetry, 2010, 21, 1563-1568.
100 “Asymmetric Deprotonation of N-Boc Piperidine: React IR Monitoring and Mechanistic Aspects”
D. Stead, G. Carbone, P. O’Brien, K. R. Campos, I. Coldham and A. Sanderson, J. Am. Chem. Soc., 2010, 132, 7260-7261.
99 “Unexpected products from the attempted organolithium-mediated conversion of cyclic β-methoxy aziridines into allylic amines”
S. C. Coote and P.O’Brien, Tetrahedron Lett., 2010, 51, 588-590.
98 “Kinetic Resolution of P-Stereogenic Phosphine Boranes via Deprotonation using s-Butyllithium/(–)-sparteine”
J. Granander, F. Secci, P. O’Brien and B. Kelly, Tetrahedron: Asymmetry, 2009, 20, 2432-2434.
97 “Catalytic Asymmetric Deprotonation of a Phosphine Borane: Comparison of Two-ligand and One-ligand Catalysis”
S. J. Canipa, P. O’Brien and S. Taylor, Tetrahedron: Asymmetry, 2009, 20, 2407-2412.
96 “Regioselective Lithiation of Silyl Phosphine Sulfides: Asymmetric Synthesis of P-Stereogenic Compounds”
J. J. Gammon, P. O’Brien and B. Kelly, Org. Lett., 2009, 11, 5022-5025.
95 “Catalytic Asymmetric Synthesis of Piperidines from Pyrrolidine: Concise Synthesis of (+)-L-733,060”
J. L. Bilke, S. P. Moore, P. O’Brien and J. Gilday, Org. Lett., 2009, 11, 1935-1938.
94 “Lithiation-Electrophilic Trapping of N-Sulfonyl-activated Ethylene Aziridines”
J. Huang, S. P Moore, P. O’Brien, A. C Whitwood and J. Gilday, Org. Biomol. Chem., 2009, 7, 335.
93 “Stereoselective Aziridination of Cyclic Allylic Alcohols Using Chloramine-T”
S. C. Coote, P. O’Brien and A. C. Whitwood, Org. Biomol. Chem., 2008, 6, 4299-4314.
92 “Organolithium-mediated Conversion of b-Alkoxy Aziridines into Allylic Sulfonamides: Effect of the N-Sulfonyl Group and a Formal Synthesis of (±)-Perhydrohistrionicotoxin”
S. C. Coote, S. P. Moore, P. O’Brien, A. C. Whitwood and J. Gilday, J. Org. Chem., 2008, 73, 7852-7855.
91 “On the Two-Ligand Catalytic Asymmetric Deprotonation of N-Boc Pyrrolidine: Probing the Effect of the Stoichiometric Ligand”
J. L. Bilke and P.O’Brien, J. Org. Chem., 2008, 73, 6452-6454.
90 “Catalytic Asymmetric Deprotonation of Phosphine Boranes and Sulfides as a Route to P-Stereogenic Compounds”
J. J. Gammon, S. J. Canipa, P. O’Brien, B. Kelly and S. Taylor, Chem. Commun. 2008, 3750-3752.
89 “Palladium-Catalyzed Enantioselective Oxidation of Chiral Secondary Alcohols: Access to Both Enantiomeric Series”
D. C. Ebner, R. M. Trend, C. Genet, M. J. McGrath, P. O'Brien and B. M. Stoltz, Angew. Chem. Int. Ed. 2008, 47, 6367-6370.
88 “Expanding the Synthetic Potential of Asymmetric Deprotonation: Arylation of Carbanions”
P. O’Brien and J. L. Bilke, Angew. Chem. Int. Ed. 2008, 47, 2734-2736.
87 “Basic Instinct: Design, Synthesis and Evaluation of (+)-Sparteine Surrogates for Asymmetric Synthesis”
P. O’Brien, Chem. Commun., 2008, 655-667. (Feature Article)
86 “A New Sparteine Surrogate for Asymmetric Deprotonation of N-Boc Pyrrolidine”
D. Stead, P. O’Brien and A. Sanderson, Org. Lett. 2008, 10, 1409-1412.
85 “Synthesis of Substituted Allylic Sulfonamides from b-Alkoxy Aziridines and Organolithium Reagents”
S. P. Moore, P. O’Brien, A. C. Whitwood and J. Gilday, Synlett 2008, 237-241.
84 “Stereocontrolled Synthesis and Alkylation of Cyclic b-Amino Esters: Asymmetric Synthesis of a (–)-Sparteine Surrogate”
J-P. R. Hermet, A. Viterisi, J. M. Wright, M. J. McGrath, P. O’Brien, A. C. Whitwood and J. Gilday, Org. Biomol. Chem., 2007, 5, 3614-3622.
83 “Total synthesis of the lupin alkaloid cytisine: comparison of synthetic strategies and routes”
D. Stead and P. O’Brien, Tetrahedron, 2007, 63, 1885-1897.
82 “Asymmetric deprotonation of N-Boc piperidines”
I. Coldham, P. O’Brien, J. J. Patel, S. Raimbault, A. J. Sanderson, D. Stead and D. T. E. Whittaker, Tetrahedron: Asymmetry, 2007, 18, 2113-2119.
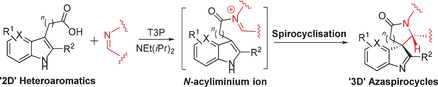
81 “New route to azaspirocycles via the organolithium-mediated conversion of b-alkoxy aziridines into cyclopentenyl amines”
S. P. Moore, S. C. Coote, P. O’Brien and J. Gilday, Org. Lett. 2006, 8, 5145-5148.
80 “Synthesis of (+)-(1R,2S,9S)-11-Methyl-7,11-Diazatricyclo[7.3.1.02,7]tridecane, a (+)-Sparteine Surrogate”
A. J. Dixon, M. J. McGrath and P. O’Brien, Org. Synth., 2006, 83, 141-154.
79 “Catalytic Asymmetric Synthesis of Ferrocenes and P-Stereogenic Bisphosphines”
C. Genet, S. J. Canipa, P. O’Brien and S. Taylor, J. Am. Chem. Soc., 2006, 128, 9336-9337.
78 “Development of a Catalytic Asymmetric Variant of Hoppe’s O-Alkyl Carbamate Deprotonation Methodology”
M. J. McGrath and P. O’Brien, Synthesis, 2006, 2233-2241.
77 “Reactivity series for s-BuLi/diamine-mediated lithiation of N-Boc pyrrolidine: applications in catalysis and lithiation of N-Boc piperidine”
M. J. McGrath, J. L. Bilke and P. O’Brien, Chem. Commun., 2006, 2607-2609.
76 “Evaluation of sparteine-like chiral diamines in the enantioselective lithiation-electrophilic trapping of an O-alkyl carbamate”
C. Genet, M. J. McGrath and P. O’Brien, Org. Biomol. Chem., 2006, 4, 1376-1382.
75 “Two-step Synthesis of N-Sulfonyl Aziridines from Epoxides”
J. Huang and P. O’Brien, Synthesis, 2006, 425-434.
74 “Catalytic Asymmetric Deprotonation Using a Ligand Exchange Approach”
M. J. McGrath and P. O’Brien, J. Am. Chem. Soc., 2005, 127, 16378-16379.
73 “Scope and limitations of the catalytic asymmetric rearrangement of epoxides to allylic alcohols using chiral lithium amide bases/lithiated imidazoles”
S. J. Oxenford, J. M. Wright, P. O’Brien, N. Panday and M. R. Shipton, Tetrahedron Lett., 2005, 46, 8315-8318.
72 “Organolithium-mediated conversion of b-functionalised aziridines into alkynyl amino alcohols and diamines”
J. Huang and P. O’Brien, Chem. Commun., 2005, 5696-5698.
71 “Concise Synthesis (±)-Cytisine via Lithiation of N-Boc Bispidine”
D. Stead, P. O’Brien and A. J. Sanderson, Org. Lett., 2005, 7, 4459-4462.
70 “Two-step syntheses of 2,4,6-triisopropylbenzenesulfonyl aziridines”
J. Huang and P. O’Brien, Tetrahedron Lett., 2005, 46, 3253-3256.
69 “Reductive Alkylation of b-Alkoxy Aziridines: New Route to Substituted Allylic Amines”
C. M. Rosser, S. C. Coote, J. P. Kirby, P. O’Brien and D. Caine, Org. Lett., 2004, 6, 4817-4819.
68 “An experimental and computational study of the enantioselective lithiation of N-Boc pyrrolidine using sparteine-like chiral diamines”
P. O’Brien, K. B. Wiberg, W. F. Bailey, J.-P. R. Hermet and M. J. McGrath, J. Am. Chem. Soc., 2004, 126, 15480-15489.
67 “Chiral base route to functionalised cyclopentenyl amines: formal synthesis of the cyclopentene core of nucleoside Q”
S. J. Oxenford, P. O’Brien and M.R. Shipton, Tetrahedron Lett., 2004, 45, 9053-9055.
66 “Crystal structures of (+)-sparteine surrogate adducts of methyllithium and phenyllithium”
C. Strohmann, K. Strohfeldt, D. Schildbach, M. J. McGrath and P. O’Brien, Organometallics, 2004, 23, 5389-5391.
65 “Evaluation of (+)-Sparteine-like Diamines for Asymmetric Synthesis”
M. J. Dearden, M. J. McGrath and P. O’Brien, J. Org. Chem., 2004, 69, 5789-5792.
64 “Concise asymmetric synthesis of (–)-sparteine”
J-P. R. Hermet, M. J. McGrath, P. O’Brien, D. W. Porter and J. Gilday, Chem. Commun., 2004, 1830-1831.
63 “Lithium enolates from a (–)-quinic acid-derived cyclohexanone with a b-alkoxy leaving group: regioselective preparation and evaluation of enolate stability towards b-elimination”
L. M. Murray, P. O’Brien, R. J. K. Taylor and S. Wünnemann, Tetrahedron Lett., 2004, 45, 2596-2601.
62 “cis- and trans-Stereoselective Epoxidation of N-Protected 2-Cyclohexen-1-yl Amines”
P. O’Brien, A. C. Childs, G. J. Ensor, C. L. Hill, J. P. Kirby, M. J. Dearden, S. J. Oxenford and C. M. Rosser, Org. Lett., 2003, 5, 4955-4957.
61 “On the a-lithiation-rearrangement of N-toluenesulfonyl aziridines: mechanistic and synthetic aspects”
P. O’Brien, C. M. Rosser and D. Caine, Tetrahedron, 2003, 59, 9779-9791.
60 “Synthesis of sparteine-like chiral diamines and evaluation in the enantioselective lithiation-substitution of N-(tert-butoxycarbonyl)pyrrolidine”
J-P. R. Hermet, D. W. Porter, M. J. Dearden, J. R. Harrison, T. Koplin, P. O’Brien, J. Parmene, V. Tyurin, A. C. Whitwood, J. Gilday and N. M. Smith, Org. Biomol. Chem., 2003, 1, 3977-3988.
59 “a-Lithiation-rearrangement of N-toluenesulfonyl aziridines with sec-butyllithium and (–)-sparteine: opposite sense of asymmetric induction to epoxides”
P. O’Brien, C. M. Rosser and D. Caine, Tetrahedron Lett., 2003, 44, 6613-6615.
58 “Stereoselective Reactions of a (–)-Quinic Acid-Derived Enone: Application to the Synthesis of the Core of Scyphostatin”
L. M. Murray, P. O’Brien and R. J. K. Taylor, Org. Lett., 2003, 5, 1943-1946.
57 “Chiral lithium amide base-mediated rearrangement of meso-cyclohexene oxides: asymmetric synthesis of amino- and aziridinocyclohexenols”
P. O’Brien and C. D. Pilgram, Org. Biomol. Chem., 2003, 1, 523-534.
56 “An azetidinium ion approach to 3-aryloxy-3-aryl-1-propanamines”
P. O’Brien, D. W. Phillips and T. D. Towers, Tetrahedron Lett., 2002, 43, 7333-7335.
55 “A Readily-Accessible (+)-Sparteine Surrogate”
M. J. Dearden, C. R. Firkin, J-P. R. Hermet and P. O’Brien, J. Am. Chem. Soc., 2002, 124, 11870-11871.
54 “Optimisation of Enantioselectivity for the Chiral Base-mediated Rearrangement of Bis-protected meso-4,5-Dihydroxycyclohexene Oxides: Asymmetric Synthesis of 4-Deoxyconduritols and Conduritol F”
S. E. de Sousa, P. O'Brien and C. D. Pilgram, Tetrahedron, 2002, 58, 4643-4654.
53 “Diastereocontrolled Synthesis of trans- and cis-meso-Cyclopentene Aziridines”
D. Caine, P. O'Brien and C. M. Rosser, Org. Lett., 2002, 4, 1923-1926.
52 “Synthesis and reactions of cyclopentadiene monoaziridine: a concise approach to the core of agelastatin A”
E. Baron, P. O'Brien and T. D. Towers, Tetrahedron Lett., 2002, 43, 723-726.
51 “Diamine Synthesis: Exploring the Regiochemistry of Ring Opening of Aziridinium Ions”
P. O'Brien and T. D. Towers, J. Org. Chem., 2002, 67, 304-307.
50 “Chiral base route to cyclic polyols: asymmetric synthesis of aminodeoxyconduritols and conduritol F”
S. E. de Sousa, P. O'Brien and C. D. Pilgram, Tetrahedron Lett., 2001, 42, 8081-8083.
49 “Asymmetric Synthesis of N-Substituted (R)-2-[(Pyrrolidin-1-yl)methyl]pyrrolidines”
J. R. Harrison and P. O'Brien, Synth. Commun., 2001, 31, 1155-1160.
48 “The synthesis and spectroscopic characterisation of chiral meso-tetraarylmetalloporphyrins bearing meso-pentafluorophenyl groups”
S. P. Foxon, J. R. Lindsay Smith, P. O'Brien and G. Reginato, J. Chem. Soc., Perkin Trans. 2, 2001, 1145-1153.
47 “Evaluation of a sparteine-like diamine for asymmetric synthesis”
J. R. Harrison, P. O'Brien, D. W. Porter and N. M. Smith, Chem. Commun., 2001, 1202-1203.
46 “Practical One-Step Synthesis of Koga's Chiral Bases”
E. Cuthbertson, P. O'Brien and T. D. Towers, Synthesis, 2001, 693-695.
45 “Stoichiometric asymmetric processes”
P. O'Brien, J. Chem. Soc., Perkin Trans. 1, 2001, 95-113.
44 “New route to 4-aminocyclopenten-2-en-1-ols: synthesis and enantioselective rearrangement of 4-amino substituted cyclopentene oxides”
S. Barrett, P. O'Brien, H. C. Steffens, T. D. Towers and M. Voith, Tetrahedron, 2000, 56, 9633-9640.
43 “Asymmetric Synthesis of Cyclic b-Amino Acids”
P. O'Brien, D. W. Porter and N. M. Smith, Synlett, 2000, 1336-1338.
42 “Bispidine-derived N-acyliminium ions in synthesis: stereocontrolled construction of the BCD rings of sparteine”
J. R. Harrison and P. O'Brien, Tetrahedron Lett., 2000, 41, 6167-6170.
41 “Diastereoselective synthesis of sparteine analogues via lithiation-electrophilic quench of N-Boc bispidines”
J. R. Harrison and P. O'Brien, Tetrahedron Lett., 2000, 41, 6161-6165.
40 “Diastereoselective epoxide rearrangements using lithium amide bases: first stereocontrolled synthesis of 4-deoxyconduritols”
A. Kee, P. O'Brien, C. D. Pilgram and S. T. Watson, Chem. Commun., 2000, 1521-1522.
39 “Studies towards the preparation of sparteine-like diamines for asymmetric synthesis”
J. R. Harrison, P. O'Brien, D. W. Porter and N. M. Smith, J. Chem. Soc., Perkin Trans. 1, 1999, 3623-3632.
38 “Enantioselective rearrangement of a meso-cyclohexene oxide using norephedrine-derived chiral bases”
B. Colman, S. E. de Sousa, P. O'Brien, T. D. Towers and W. Watson, Tetrahedron: Asymmetry, 1999, 10, 4175-4182.
37 “Synthesis and Enantioselective Rearrangement of meso-Aziridino Cyclohexene Oxides”
P. O'Brien and C. D. Pilgram, Tetrahedron Lett., 1999, 40, 8427-8430.
36 “A New Norephedrine-derived Chiral Base for Epoxide Rearrangement Reactions”
S. E. de Sousa, P. O'Brien and H. C. Steffens, Tetrahedron Lett., 1999, 40, 8423-8425.
35 “An efficient protocol for a Sharpless-style racemic dihydroxylation”
J. Eames, H. J. Mitchell, A. Nelson, P. O'Brien, S. Warren and P. Wyatt, J. Chem. Soc., Perkin Trans. 1, 1999, 1095-1103.
34 “Chiral base-mediated benzylic functionalisation of (alkyl benzyl ether)tricarbonylchromium(0) complexes: a structure-reactivity study”
S. E. Gibson (née Thomas), P. O'Brien, E. Rahimian and M. H. Smith, J. Chem. Soc., Perkin Trans. 1, 1999, 909-912.
33 “Asymmetric Aminohydroxylation: Scope, Limitations and Use in Synthesis”
P. O'Brien, Angew. Chem., Int. Ed. Engl., 1999, 38, 326-329.
32 “Stereoselective Epoxidation of Cyclic Alkenes Using m-CPBA and Oxone®/Trifluoroacetone – a Comparison”
S. E. de Sousa, P. O'Brien, C. D. Pilgram, D. Roder and T. D. Towers, Tetrahedron Lett., 1999, 40, 391-392.
31 “Unexpected Effect of Protecting Group and Solvent on the Stereoselectivity of m-CPBA Epoxidation of Diprotected cis-4,5-Dihydroxycyclohexenes”
S. E. de Sousa, A. Kee, P. O'Brien and S. T. Watson, Tetrahedron Lett., 1999, 40, 387-390.
30 “Mechanistic and synthetic aspects of stereoselective reactions of lithium derivatives of chiral phosphine oxides: X-ray crystal structure of (1R*,2S*,1'S*,2'R*)-1-(1'-diphenylphosphinoyl-2'-phenylpropyl)-2-phenylsulfanylcyclohexan-1-ol”
C. Guéguen, P. O'Brien, H. R. Powell, P. R. Raithby and S. Warren, J. Chem. Soc., Perkin Trans. 1, 1998, 3405-3417.
29 “Synthesis and Enantioselective Rearrangement of 4-Amino-substituted Cyclopentene Oxides”
P. O'Brien, T. D. Towers and M. Voith, Tetrahedron Lett., 1998, 39, 8175-8178.
28 “Asymmetric aminohydroxylation of substituted styrenes: applications in the synthesis of enantiomerically enriched arylglycinols and a diamine”
P. O'Brien, S. A. Osborne and D. D. Parker, J. Chem. Soc., Perkin Trans. 1, 1998, 2519-2526.
27 “Chiral lithium amide base-mediated rearrangement of diprotected meso 4,5-dihydroxy cyclohexene oxides: enantioselective synthesis of (4R,5S)- and (4S,5R)-4,5-di(tert-butyldimethylsilyloxy)cyclohex-2-enone”
P. O'Brien and P. Poumellec, J. Chem. Soc., Perkin Trans. 1, 1998, 2435-2441.
26 “Two expedient methods for the preparation of chiral diamines”
S. E. de Sousa, P. O'Brien and P. Poumellec, J. Chem. Soc., Perkin Trans. 1, 1998, 1483-1492.
25 “Recent advances in the use of chiral lithium amide bases in synthesis”
P. O'Brien, J. Chem. Soc., Perkin Trans. 1, 1998, 1439-1457.
24 “Asymmetric Aminohydroxylation of Substituted Styrenes Using t-Butyl Carbamate”
P. O'Brien, S. A. Osborne and D. D. Parker, Tetrahedron Lett., 1998, 39, 4099-4102.
23 “Stereocontrolled Synthesis of Functionalised Cyclohexanes via the Lithium Amide-mediated Rearrangement of a meso-4,5-Disubstituted Cyclohexene Oxide”
P. O'Brien and J. J. Tournayre, Tetrahedron, 1997, 53, 17527-17542.
22 “Two Step Synthesis of C2 Symmetric 2,3-Diarylalkyloxybenzaldehydes – A Mitsunobu Approach”
J. R. Lindsay Smith, P. O'Brien and G. Reginato, Tetrahedron: Asymmetry, 1997, 8, 3415-3420.
21 “Two useful methods for the preparation of (R)- and (S)-N-methyl-1-phenyl-2-(1-pyrrolidinyl)ethanamine”
S. E. de Sousa, P. O'Brien and P. Poumellec, Tetrahedron: Asymmetry, 1997, 8, 2613-2618.
20 “Aziridinium Ions from Phenylglycinol – A New Approach to the Synthesis of Chiral Diamines”
S. E. de Sousa and P. O'Brien, Tetrahedron Lett., 1997, 38, 4885-4888.
19 “Cyclobutanone as an electrophile for lithiated phosphine oxides: “Internal Quench” Conditions”
C. Guéguen, P. O'Brien, S. Warren and P. Wyatt, J. Organomet. Chem., 1997, 529, 279-283.
18 “Experimental and Molecular Orbital Calculational Study of the Stereoselective Horner-Wittig Reaction with Phosphine Oxides: Control of Stereoselectivity by Lithium”
D. R. Armstrong, D. Barr, M. G. Davidson, G. Hutton, P. O'Brien, R. Snaith and S. Warren, J. Organomet. Chem., 1997, 529, 29-33.
17 “Investigation of the configurational stability of lithiated phosphine oxides using the Hoffmann test: X-ray structures of (2R*,3S*,4S*)-4-(N,N-dibenzylamino)-2-diphenylphosphinoyl-5-phenylpentan-3-ol and (2S*,4S*)-4-(N,N-dibenzylamino)-2-diphenylphosphinoyl-5-phenylpentan-3-one”
P. O'Brien, H. R. Powell, P. R. Raithby and S. Warren, J. Chem. Soc., Perkin Trans. 1, 1997, 1031-1039.
16 “Asymmetric Synthesis with Diphenylphosphine Oxides: Bicyclic Aminals and Oxazolidines as Chiral Auxiliaries”
P. O'Brien and S. Warren, Tetrahedron: Asymmetry, 1996, 7, 3431-3444.
15 “Investigation of the configurational stability of lithiated phosphine oxides using diastereomerically pure and enantiomerically enriched phosphine oxides”
P. O'Brien and S. Warren, J. Chem. Soc., Perkin Trans. 1, 1996, 2567-2573.
14 “Chiral Base-mediated Rearrangement of meso-Cyclohexene Oxides to Allylic Alcohols”
P. O'Brien and P. Poumellec, Tetrahedron Lett., 1996, 37, 8057-8058.
13 “Chiral Phosphine Oxides and Chiral Esters in Stereoselective Intermolecular Acylation Reactions of Phosphine Oxides”
D. Cavalla, C. Guéguen, A. Nelson, P. O'Brien, M. G. Russell and S. Warren, Tetrahedron Lett., 1996, 37, 7465-7468.
12 “Highly Stereoselective Hydroxy-alkylation, Silylation and Alkylation Reactions of Lithium Derivatives of Chiral Phosphine Oxides”
C. Guéguen, H. J. Mitchell, P. O'Brien and S. Warren, Tetrahedron Lett., 1996, 37, 7461-7464.
11 “Amino Acid Derived Thiane Oxide and Dioxide Systems as Disposable Templates: Synthesis of a-Amino Ketones, anti amino alcohols and an Amino Cyclopentenone”
M. P. Gamble, G. M. P. Giblin, J. G. Montana, P. O'Brien, T. P. Ockenden and R. J. K. Taylor, Tetrahedron Lett., 1996, 37, 7457-7460.
10 “Synthesis of R or S diphenylphosphinoyl hydroxy aldehydes and 1,2 diols using Mukaiyama's bicyclic aminal methodology and Sharpless asymmetric dihydroxylation”
P. O'Brien and S. Warren, J. Chem. Soc., Perkin Trans. 1, 1996, 2129-2138.
9 “Stereocontrolled route to some optically active b-hydroxy phosphine oxides using the stereoselective addition of metallated phosphine oxides to proline-derived keto aminals”
P. O'Brien and S. Warren, J. Chem. Soc., Perkin Trans. 1, 1996, 2117-2127.
8 “A Simple and Efficient Method for the Preparation of Homochiral Amines: Application to the Synthesis of a New C2 Symmetric Triamine”
P. O'Brien and P. Poumellec, Tetrahedron Lett., 1996, 37, 5619-5622.
7 “Cesium Fluoride-mediated Horner-Wittig Addition Reactions of Silyl Phosphine Oxides; Synthesis of Optically Active Silyl Phosphine Oxides Using Chiral Bases and a Chiral Acid”
P. O'Brien and S. Warren, Synlett, 1996, 579-581.
6 “Synthesis of Phenylalanine-derived b-Hydroxy and b-Keto Phosphine Oxides – Investigation of the Configurational Stability of Lithiated Phosphine Oxides Using the Hoffmann Test”
P. O'Brien and S. Warren, Tetrahedron Lett., 1996, 37, 4271-4274.
5 “Norephedrine-derived Oxazolidines as Chiral Auxiliaries – Stereocontrolled Routes to R or S b-Hydroxy Phosphine Oxides”
P. O'Brien and S. Warren, Tetrahedron Lett., 1996, 37, 3051-3054.
4 “On the Configurational Stability of Lithiated Phosphine Oxides”
P. O'Brien and S. Warren, Tetrahedron Lett., 1995, 36, 8473-8476.
3 “Asymmetric Dihydroxylations of Allylic Phosphine Oxides”
A. Nelson, P. O'Brien and S. Warren, Tetrahedron Lett., 1995, 36, 2685-2688.
2 “Stereocontrolled Synthesis of R or S Diphenylphosphinoyl Hydroxy Aldehydes and 1,2 Diols Using Bicyclic Aminals”
P. O'Brien and S. Warren, Tetrahedron Lett., 1995, 36, 2681-2684.
1 “An Efficient Protocol for a Sharpless Style Racemic Dihydroxylation”
J. Eames, H. J. Mitchell, A. Nelson, P. O'Brien, S. Warren and P. Wyatt, Tetrahedron Lett., 1995, 36, 1719-1722.
Group Members
Current Group Members
PhD Students
Andres Gomez-Angel

Andres completed his chemistry and chemical engineering bachelor studies at Los Andes University in Colombia. He then undertook a MSc by research in the O'Brien group on the initial phase of the 3-D building block project. He has since returned to the O’Brien group for his PhD studies, supported by the Wild Fund. His project focuses on the development of 3-D building blocks for fragment elaboration in medicinal chemistry. His hobbies include playing tenor saxophone, building and repairing computers for his friends and cooking overly complicated meals.
Lucy Tomczyk

Lucy obtained her Mchem degree from the University of York, spending the final year in the O’Brien group working on a new synthetic route to (–)-quinine. She then returned to York to begin a Ph.D. (EPSRC iCASE), supervised by Professor Peter O’Brien and Professor Ian Fairlamb, focusing on the stereospecific C(sp3)-C(sp2) Suzuki-Miyaura cross-coupling of heterocyclic boronates. In her spare time Lucy enjoys sewing, baking and playing card games with friends.
Stuart McHale

Stuart completed his MChem (with medicinal chemistry) degree at Newcastle University (2017-2021) working in the research project group of Dr Kate Madden investigating small molecule analogues of berberine as potential therapeutics in neurodegenerative diseases. He also carried out an industrial placement (2019-2020) with the Centre for Process Innovation (CPI) working on in-line analysis for continuous twin-screw wet granulation. In 2021 he took a PhD position co-supervised by Peter O’Brien and Ian Fairlamb and is currently working on stereospecific C(sp3)-C(sp2) Suzuki-Miyaura cross-couplings of saturated heterocyclic boronates.
Stuart Smith

Stuart obtained an Integrated Masters degree in Chemistry with Medicinal Chemistry from the University of Glasgow in 2021. He completed several research placements during this time (mainly in molecular genetics and chemical automation), as well as a placement year at GlaxoSmithKline. In October 2021, he joined the Department of chemistry at the University of York to complete a PhD under the supervision of Professors Ian Fairlamb and Peter O'Brien. His work involves automating exploration of the chemical space of C(sp3)-C(sp2) Suzuki-Miyaura cross-couplings, in conjunction with statistical and machine learning approaches, to facilitate reaction development and aid mechanistic understanding of this challenging reaction for industrial pharmaceutical applications.
Xinyu Wang

Xinyu obtained his bachelor and Master's degree in China. He worked as a synthetic chemist in the CRO industry for 5 years. He joined the O'Brien group in 2022 under the support of CSC (China Scholarship Council). His project focuses on the fragment-based lead generation of small molecule inhibitors of the Nsp3 macrodomain in SARS-CoV-2.
Will Butler

Will obtained his MChem degree from the University of York, spending the final year in the O’Brien group developing fragment-based small molecule inhibitors of the Nsp3 Macrodomain in SARS-CoV-2. Will then returned to York to undertake a Ph.D., supervised by Professor Peter O’Brien, focusing on new 3-D building blocks for fragment elaboration in medicinal chemistry, as well as returning to the aforementioned Nsp3 Macrodomain project. Outside of the lab, Will enjoys running, bouldering, photography and a good pint.
Yuran Wang

Yuran completed his BSc in chemistry and MRes in Organic chemistry: Drug discovery at University College London, investigating novel inhibitors of the SARS-CoV-2 main protease. In January 2023, he joined the O’Brien Group for his PhD. His project is on 3-D fragment elaboration in conjunction with fragment-based drug discovery of inhibitors of the SARS-CoV-2 Nsp3 Macrodomain. In his free time, Yuran enjoys ice skating, video games, and board games such as weiqi and chess.
Masters students
Islam Araar

Islam obtained a 1st Class Honours degree from Kingston University in Pharmaceutical Sciences and was awarded for top performance. Research was conducted under the supervision of Dr. Stephen Wren in his final year developing synthetic methodologies for accessing bioactive fluorenes. In 2022, Islam joined the O’Brien group as a MSc by Research student, working on the elaboration of fragment hits in 3D to produce medicinally relevant compounds. His academic interests include synthetic organic chemistry, process chemistry, natural products and pharmaceutics. Outside of the lab, Islam enjoys doing many sports including weight lifting, long/short distance running and Judo.