Synthesis of Nitrogen and Oxygen Heterocycles using Organolithium Chemistry
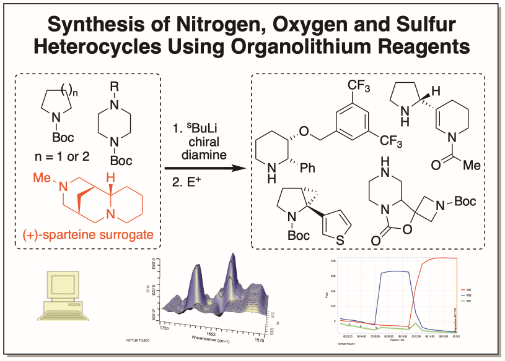
The O’Brien group carries out a distinctive, multi-disciplinary approach to mechanistic interrogation of organolithium/diamine-mediated reactions. We combine several orthogonal techniques including in situ React IR spectroscopy (for exploring lithiation and trapping reactions in real-time), NMR spectroscopy (for determining the solution structure of organolithium/diamine complexes), DFT computational modelling and more traditional reaction optimisation (e.g. solvent and temperature variation). All of this information is then utilised in optimising reaction conditions/yields and in the development of new methodology, with a focus on high levels of enantioselectivity, diastereoselectivity, regioselectivity and chemoselectivity. Then, our new methods are applied to the synthesis of nitrogen and oxygen heterocycles such as pyrrolidines, piperidines, piperazines and morpholines, which are amongst the most common structural motifs in blockbuster pharmaceuticals. As a result, the products we generate are of interest to medicinal chemistry groups and, since 2010, together with EPSRC funding, this area of research has benefitted from industrial support from AstraZeneca, GlaxoSmithKline, Merck and Pfizer.
