Dr Anne Routledge
01904 324540
Email: anne.routledge@york.ac.uk
Bio-organic/solid-phase chemistry
We have a variety of research themes in the area bio-organic/synthetic chemistry, key themes are outlined below
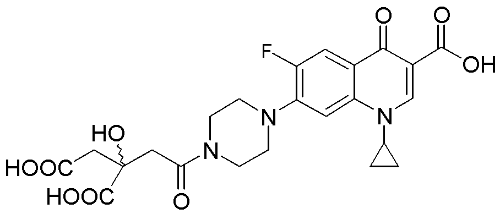
Fluoroquinolone analogs to target bacterial resistance
Fluoroquinolone analogs are being synthesized to target increasingly resistant strains of bacteria. We aim to increase the intracellular drug concentration and improve efficacy by overcoming resistance arising from changes to cell permeability and drug efflux. The synthesised analogs are designed to increase efficacy by facilitating active transport through the bacterial cell membranes and preventing efflux. We aim to achieve this by exploiting bacterial nutrient uptake mechanisms (an example is shown below).
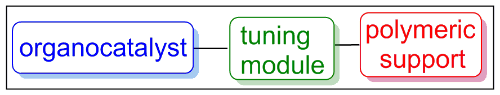
Development of ‘tuning modules’ to enhance the green credentials of heterogeneous organocatalysis
Organocatalysis, the catalysis of reactions using small organic molecules, fulfils some green chemistry guidelines by avoiding the use of endangered metals and in being catalytic. Immobilising a catalyst on an inert polymer bead facilitates catalyst recovery and reuse which adds significant greening of the chemistry and provides a substantial cost benefit both in allowing the catalyst to be reused and simplifying the work-up procedure. The third aspect of organocatalysis that needs be greened is the ability to efficiently perform the reaction in a sustainable solvent whilst using an immobilised catalyst. We are addressing this by developing tuning modules (see below). Our goal is to have a set of tuning modules for different solvent, catalyst and polymer support combinations.
Design and application of novel analytical constructs to facilitate MS proteome analysis.
Since the sequencing of the human genome attention has now turned to the discipline of proteomics which explores the nature and levels of proteins within cells. Although the genome is complex, the human proteome is vastly more complex, containing around a million proteins. Biologically interesting protein samples tend not only to be highly complex, but often available in only in very limited amounts, so that the dual challenges of sample complexity and limited sample quantities are inherent in proteomic studies.
Our research aims to provide chemical tools to facilitate mass spectrometric analysis of peptides resulting from proteolytic digestion of protein samples.
Selected References
A Salmochelin S4-Inspired Ciprofloxacin Trojan Horse Conjugate, T.J. Sanderson, C.M. Black, J.W. Southwell, E.J. Wilde, A. Pandey, R. Herman, G.H. Thomas, E. Boroz, A.K. Duhme-Klair and A. Routledge, ACS Infect. Dis. 2020, 6(9), 2532-2541.
Investigation of Parameters that Affect Resin Swelling in Green Solvents, C.Amadi‐Kamalu, H. Clarke, M. McRobie, J. Mortimer, M. North, Y. Ran, A. Routledge, D. Sibbald, M. Tickias, K. Tse and H. Willway, ChemistryOpen, 2020, 9(4), 431-441.
Mimicking salmochelin S1 and the interactions of its Fe(III) complex with periplasmic iron siderophore binding proteins CeuE and VctP, E.J. Wilde, E.V. Blagova, T.J. Sanderson, D.J. Raines, R.P. Thomas, A. Routledge, A.K. Duhme-Klair and K.S. Wilson, J. Inorg. Biochem., 2019, 190, 75-84.
Perspectives for New Anti-infectives Targeting Iron Uptake Systems, U. Bilitewski, J. A. V. Blodgett, A.K. Duhme-Klair, S. Dallavalle, S. Laschat, A. Routledge, and R. Schobert, Angew. Chem. Int. Ed., 2017, 56, 14360-14382.
Greener Solvents for Solid-Phase Synthesis, S. B. Lawrenson, M. North, F. Peigneguy and A. Routledge, Green Chem., 2017, 19, 952–962.

