Professor Caroline Dessent
+44 (0)1904 324092
Email: caroline.dessent@york.ac.uk
Biography
Caroline grew up in Gilfach Goch, South Wales, and attended Tonyrefail Comprehensive School. She studied Chemistry at Jesus College Oxford, where her tutors included Prof Mike Pilling and Prof Anthony Downs. She spent her Part II researching atmospheric chemistry in Prof Richard Wayne’s group in the Physical and Theoretical Chemistry Laboratory. Caroline studied for her PhD at Yale University under the supervision of Prof Mark Johnson. Her thesis work was on laser spectroscopic studies of the electronic excited states of negative ions and free electron-molecule interactions. Her thesis was awarded the Richard Wolfson prize.
Following her PhD, she came to York as a post-doctoral researcher to work with Prof Klaus Muller-Dethlefs on photoionization spectroscopy of molecules and clusters. She was a Royal Society University Research Fellow from 1999-2008, and was then awarded a prestigious European Research Council Starting Independent Research Award in the inaugural ERC funding round. Her ERC grant, BIOIONS, focused on the development of a new instrument and spectroscopic techniques for studying biological ions in the gas-phase. Following her ERC grant, she was promoted to Reader (2017) and then Professor of Physical Chemistry (2020).
Caroline’s research group uses the novel instrumentation developed through her ERC grant, laser-interfaced mass spectrometers, to study the properties of a wide range of photoactive molecular systems that are of interest in biology and human health. Current areas of study include organic sunscreens, photopharmaceuticals, and photolytic breakdown pathways of emerging pollutants in the natural environment.
She is the current Chair of the Royal Society of Chemistry Spectroscopy and Dynamics Group (2019-2022), and is an elected member of the RSC Faraday Council (2021-2024). She has also been an active member of the Institute of Physics, having served in a number of roles for the Molecular Physics Group.
Caroline has combined her scientific career with raising three children (James, Grace and Kitty), and worked part time between 2009 and 2020. She is a passionate supporter of Equality, Diversity and Inclusion, and was previously the Chair of the Department’s Equality & Diversity Group, as well as the Science Faculty’s Athena Swan Steering Group. She led the Chemistry Department’s successful 2018 Gold Athena Swan submission. Caroline is strongly committed to improving the participation of minority ethnic scientists in Academia. She established work in the Department to Decolonize the Chemistry Curriculum, and runs a project to investigate the lived experienced of minoritised Chemistry students and staff.
Research
Photoactive Molecules and Ensembles;
Laser-Interfaced Mass Spectrometry
My research interests lie in understanding the fundamental properties of a broad range of photoactive molecules and ensembles. In particular, my focus is on photoactive biological molecules, as well as molecular systems where their photochemical properties can be applied to health problems. My research group employs a novel combination of optical laser spectroscopy along with advanced mass spectrometry techniques. We apply these methods to obtain robust photochemical and photophysical information by examining the systems in the highly-controllable gas-phase environment.
Inorganic Metal Complex-Biomolecule Aggregates to Model Photopharmaceuticals
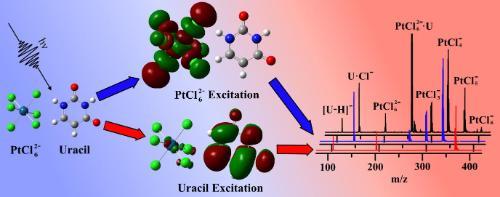
Photoactivated chemotherapy (PACT) offers both temporal and spatial control over drug activation and has considerable potential for future treatments of cancer. We are investigating metal complex-nucleobase clusters as model systems for identifying fundamental photochemical processes occurring in PACT anti-cancer platinum drug therapies that target DNA. For example, isolated molecular clusters of adenine, cytosine, thymine and uracil bound to hexachloroplatinate, PtCl62-, have been studied using laser electronic photodissociation spectroscopy to investigate photoactivation of a platinum complex in the vicinity of a nucleobase. This is the first study to explore the specific role of a strongly photoactive platinum compound within an aggregate complex. Our results demonstrate how wavelength-dependent selective photoexcitation can drive distinctive photo decay channels (eg electron transfer from the metal complex to the nucleobase or proton-abstraction from the nucleobase) for a model photo-pharmaceutical. Such studies provide critical benchmarking data for developing QM/MM methodologies for modelling photoactive metal complex-biomolecule interactions and have the potential to provide a route for facile screening of possible photopharmaceuticals. Current work involves extending these studies to metal-complex-oligonucleotide ensembles.
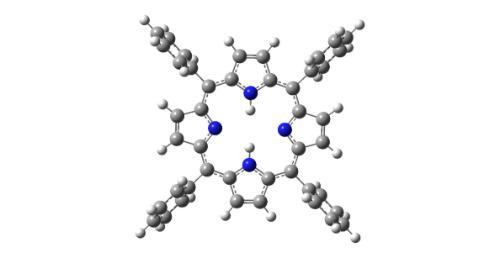
Porphyrins and Metalloporphyrins: Structure versus Function
There is intense current interest in the photophysical properties of porphyrins and metalloporphyrins (MPs) due to their potential applications in many key chemical areas including photomedicine and optoelectronic materials. MPs are common molecular structures in nature; hemoglobins and myoglobins contain MPs as the active centres, while oxovanadium and nickel alkyl porphyrins are widely distributed in fossil fuels. We are currently studying how the structural features of isolated porphyrins and metalloporphyrins influence their intrinsic electronic spectra, to provide an improved fundamental understanding of the nature of the excited states. Further experiments will focus on metalloporphyrin-aromatic molecule clusters to probe the effect of direct axial coordination to the metal centre on the MP electronic spectrum. A fundamental understanding of the nature of porphyrin and metalloporphyrin excited states is essential to inform the future rational design of new porphyrin materials.
Organic Sunscreen Photodegradation
Melanoma is one of the most common cancers in the UK, with the number of reported cases increasing annually. As a result, considerable efforts have been made towards developing more efficient broad spectrum sunscreens that protect against UVA and UVB. Given the importance of sunscreens to human health, it is surprising that relatively little is known at the molecular level about how photoactive sunscreen molecules absorb and disperse the damaging UV radiation into less harmful forms of energy. We are performing a new series of experiments using laser-interfaced mass spectrometry to study the photodegradation routes of organic sunscreens. One example, relates to how the pH environment of a sunscreen molecule affects its photostability.
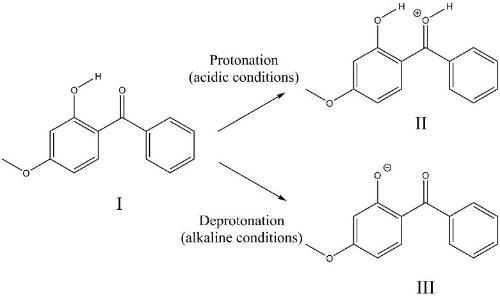
The effect of pH on the sunscreen, oxybenzone (I). How does this affect photostability?
We are developing new techniques to link solution-phase photochemistry of sunscreen molecules (via real-time sampling) to their intrinsic (i.e. in vacuo) photochemical properties.
Laser-Interfaced Mass Spectrometry
We have pioneered work to combine commercial ion trap mass spectrometers (Bruker Esquire and amaZon) with pulsed, tuneable bench-top lasers. These instruments allow us to unambiguously mass select molecular systems of interest, and then subject them to laser excitation. The subsequent photochemistry and photophysics can then be investigated using the MSn features of the mass spectrometer. These experiments provide a clear picture of the intrinsic photochemical properties of a system, away from any complications that can occur when systems are studied within the liquid phase. Current work involves i) developing IR laser-mass spectrometry techniques as a “mass-resolved FTIR” fingerprinting approach, and ii) developing new spectroscopic methods to allow mass-resolved detection of chiral molecules.
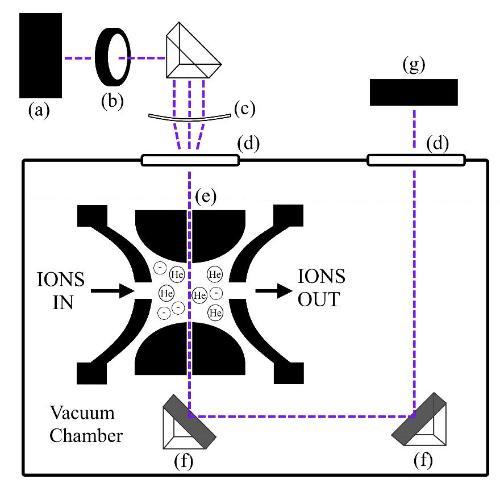
Schematic diagram of the laser-interfaced Bruker amaZon mass spectrometer.

Optimizing the laser alignment into the mass spectrometer.

