Emeritus Professor Andrzej Marek Brzozowski
01904 328265
Email: marek.brzozowski@york.ac.uk
Structural Endocrinology and Medicinal Chemistry
Research Summary
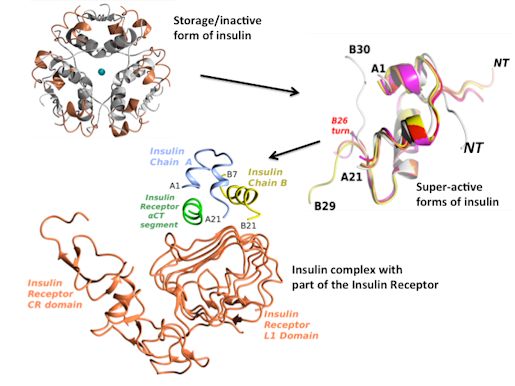
My main interest is in understanding of molecular bases of hormonal signaling that are at the cross-section of metabolism, growth (cancer) and life-span control. Human insulin, and its closely related Insulin-Like Growth Factors-I/II (IGF-I/II), governs these pathways, transducing signals through highly homologous Insulin and IGF Receptors (IR and IGF-1R). Current projects here include:
- elucidation of insulin:IR interactions via structural studies of various insulin analogues and IR ectodomain fragments (i.e. 3-D description of the so-called site 1 and site 2 in insulin:IR complex);
- development of a sustainable production of IR and IGF-1R fragments for structural studies;
- development of novel insulin analogues and its organo-mimetics for oral delivery of this hormone;
- understanding of the structural bases for metabolic-mitogenic divergence between insulin and IGFs.
I also pursue application of structural biology to facilitate progress in tackling of the neglected diseases studying structure-function relationships in proteins important for vaccine development against Leishmania sp. and Chlamydia trachomatis.
Selected publications
- Structures of insect Imp-L2 suggest an alternative strategy for regulating the bioavailability of insulin-like hormones.
Roed NK, Viola CM, Kristensen O, Schluckebier G, Norrman M, Sajid W, Wade JD, Andersen AS, Kristensen C, Ganderton TR, Turkenburg JP, De Meyts P, Brzozowski AM, Nat Commun., 2018, 9, Article 3860- Site-selective C-C modification of proteins at neutral pH using organocatalyst-mediated cross aldol ligations.
Spears RJ, Brabham RL, Budhadev D, Keenan T, McKenna S, Walton J, Brannigan JA, Brzozowski AM, Wilkinson AJ, Plevin M, Fascione MA, Chem Sci., 2018, 9, 5585-5593- Converting Insulin-like Growth Factors 1 and 2 into High-Affinity Ligands for Insulin Receptor Isoform A by the Introduction of an Evolutionarily Divergent Mutation.
Macháčková K, Chrudinová M, Radosavljević J, Potalitsyn P, Křížková K, Fábry M, Selicharová I, Collinsová M, Brzozowski AM, Žáková L1, Jiráček J, Biochemistry, 2018, 57, 2373-2382- How insulin engages its primary binding site on the insulin receptor.
J G Menting, J Whittaker, M B Margetts, L J Whittaker, G K-W Kong, B J Smith, C W Watson, L Žáková, E Kletvíková, J Jiráček, D F Steiner, S J Chan, S.J., G G Dodson, A M Brzozowski, M W Weiss, C W Ward and M C Lawrence, Nature, 2013, 493, 241-245.- Design and Synthesis of Inhibitors of Plasmodium falciparum N-myristoyltransferase, a Promising Target for Anti-Malarial Drug Discovery.
Z Yu, J Brannigan, D Moss, A M Brzozowski, A J Wilkinson, A Holder, E Tate and R J Leatherbarrow, Medicinal Chemistry, 2012, 55, 8879-8890.- Insulin and Insulin-like Growth Factor II Differentially Regulate Endocytic Sorting and Stability of Insulin Receptor Isoform A.
A Morcavallo, M Genua, A Palummo, E Kletvikova, J Jiracek, A M Brzozowski, R V Iozzo, A Belfiore and A J Morrione, J Biol Chem, 2012, 287, 11422-11436.- Non-equivalent role of inter- and intra-molecular hydrogen bonds in the insulin dimer interface.
E Antolíková, L Žáková, J P Turkenburg, C W Watson, I Hančlová, M Šanda, A Cooper, T Kraus, A M Brzozowski and J Jiráček, J Biol Chem, 2011, 286, 36968-36977.- Directed epitope delivery across the Escherichia coli outer membrane through the porin OmpF.
N G Housden, J A Wojdyla, J Korczynska, I Grishkovskaya, N Kirkpatrick, A M Brzozowski and C Kleanthous, Proc Natl Acad Sci (USA), 2010, 107, 21412-21417.
Biography
A M Brzozowski received his PhD from the University of Lódz, Poland in 1980, with post-doctoral research in Max-Planck-Institut für Experimentelle Medizin (Göttingen, Germany) and the Department of Chemistry, University of York. After further work in York as Research Fellow and Reader, he was made a Professor of the University of York in 2013. His works span a very broad field of structural biology, from biotechnology and health care enzymes, nuclear receptors to insulin-related hormonal regulation of human homeostasis and disease.


