Dr Charlotte Walker
BBSRC Discovery Fellow
Research
Research
My research is on algal biology and biotechnology. I’m particularly interested in how algae utilise inorganic carbon, through physiological mechanisms such a photosynthesis or biomineralisation. Although eukaryotic algae are responsible for 30% of global primary productivity and are key drivers of biogeochemical cycles, there are still large knowledge gaps in our understanding of the mechanisms underpinning their inorganic carbon requirements and how these are shared across lineages.
The Algal CO2 Concentrating Mechanism
The algal CO2 concentrating mechanism (CCM) is a process by which algae enhance their photosynthetic efficiency by concentrating CO2 around the photosynthetic enzyme Rubisco (Figure 1). The CCM relies heavily on HCO3- uptake from the environment and transport through the algal cell. The HCO3- is then converted to CO2 in the proximity of Rubisco by an enzyme called carbonic anhydrase. I am currently researching HCO3- channels that facilitate the CCM in different algal lineages.
My current CCM research feeds directly into an effort to engineer an algal CCM into crop plants. A functioning CCM has been modelled to increase crop yield by up to 60% and as a result directly alleviate food security issues brought on by population increase and climate change. At York this bioengineering effort is led by Professor Luke Mackinder, also a member of CNAP.
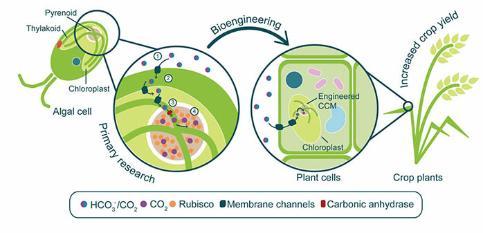
Figure 1. Research into the algal CCM is largely focused on the green alga Chlamydomonas. It is currently understood that (1) Chlamydomonas takes up inorganic carbon (Ci) from the external environment in the form of CO2 and HCO3- and (2) transports it through the cell to the thylakoid tubules in the chloroplast. (3) The HCO3- is then converted to CO2 by a carbonic anhydrase enzyme. (4) The concentrated CO2 is then delivered to the pyrenoid, the site of aggregated Rubisco, whereby it can be used to fuel photosynthesis.
Coccolithophore Calcification
Alongside carbon fixation, I’m interested in the process of algal biomineralisation, where algae make minerals to form cellular structures. My research to date has focussed on coccolithophores, these are globally abundant marine microalgae which produce ornate calcium carbonate platelets internally, these are called coccoliths. The coccoliths are then extruded to the cell surface to form a shell-like covering (Figure 2). Coccolithophores do this on such a vast scale that they are responsible for geological structures such as the White Cliffs of Dover. Their use of inorganic carbon for calcification makes them hugely influential in biogeochemistry, or global carbon cycling.

Figure 2. Scanning electron microscopy images of a range of coccolithophore species. (a) Coccolithus braarudii, (b) Scyphosphaera apsteinii, (c) Calcidiscus leptoporus, (d) Chrysotila carterae, and (e) Emiliania huxleyi. Scale bars represent 10 μm (a–d) and 1 μm (e).
Teaching and scholarship
Teaching and scholarship
![]() My aim as a teacher is to impart knowledge and enthusiasm for algal biology and biotechnology using research lead teaching. I help my students develop their fundamental biological knowledge, laboratory, data analysis and communication of research skills. As an early career researcher, I am also well positioned to support my students in their career development.
My aim as a teacher is to impart knowledge and enthusiasm for algal biology and biotechnology using research lead teaching. I help my students develop their fundamental biological knowledge, laboratory, data analysis and communication of research skills. As an early career researcher, I am also well positioned to support my students in their career development.
![]() My tutorials focus predominantly on algal biology and biotechnology. I cover a range of skills from experimental design, critical thinking, the communication of science to different audiences and research paper critiquing.
My tutorials focus predominantly on algal biology and biotechnology. I cover a range of skills from experimental design, critical thinking, the communication of science to different audiences and research paper critiquing.
![]() My projects are predominantly lab based and compliment my area of research into the algal CO2 concentrating mechanism. Students that work with me will gain a broad range of culturing techniques, molecular biology, bioinformatics, microscopy and data analysis skills. I believe a research project should promote creativity and independence and I am always willing to talk to potential students about their research ideas.
My projects are predominantly lab based and compliment my area of research into the algal CO2 concentrating mechanism. Students that work with me will gain a broad range of culturing techniques, molecular biology, bioinformatics, microscopy and data analysis skills. I believe a research project should promote creativity and independence and I am always willing to talk to potential students about their research ideas.

